Ultrasound-assisted Percutaneous Vertebroplasty in Upper and Mid-thoracic Osteoporotic Fracture
By Long Chen1, Qilin Lu1, Xiaozhen Wang1, Yiliang Zhu1, Dong Zhang2Affiliations
doi: 10.29271/jcpsp.2021.03.330ABSTRACT
Osteoporotic vertebral compression fracture (OVCF) is a common disease among the elderly individuals. With the aging of the population development, the incidence of OVCF is increasing. Cementation of vertebral body like percutaneous vertebroplasty (PVP) is a minimally invasive and effective treatment. However, the application in upper and mid-thoracic vertebrae is rare for poor fluoroscopy view caused by osteopenia vertebral body itself and scapula, sternum block. Inaccurate puncture and high radiation exposure of PVP in upper and mid-thoracic vertebrae still challenge surgeons. Owing to the ability of musculoskeletal ultrasound in revealing the clear real-time view of soft tissues and skeleton, the authors use intraoperative ultrasound-guided PVP to treat eight patients with upper or mid-thoracic vertebra body OVCFs, which has been tested to be a safe, feasible and satisfied technique after the operation and follow-up. To the authors’ knowledge, this technique has not been reported elsewhere.
Key Words: Ultrasound, Percutaneous vertebroplasty, Osteoporotic vertebral compression fracture, Upper and mid-thoracic vertebra body.
INTRODUCTION
The aging structure of social population is prominent, and the prevalence of osteoporosis is gradually increasing.1 The main complication of osteoporosis is osteoporotic vertebral compression fracture (OVCF), which can effect spinal curvature, sagittal balance and cardiopulmonary function.2 Cementation of vertebral body, including the percutaneous vertebroplasty (PVP), is a wide-used minimally invasive way for OVCF. Due to high risk and difficulty of operation, the application of PVP in upper and mid-thoracic vertebrae is rare, and the accurate puncture is a crucial procedure.3 In recent years, with the development of ultrasound technology, it has been gradually applied in spinal operation study.4,5
Ultrasound can clearly and real-time reveal soft tissues and bony parts, such as muscle, spinous and transverse process, which could assist to guide the position and direction of needle in order to improve the accuracy and safety of PVP in upper and mid-thoracic vertebrae.
TECHNIQUE
Inclusion criteria were: age 60-75 years, vertebral fracture located in T3-T8 segment, bone density T < - 2.5 (DAX method), severe back pain from fresh single segment thoracic vertebral body fracture confirmed by MRI, compression less than 1/3 of vertebral body height without lesion in posterior wall of vertebral body, and informed consent for surgery and cooperation in postoperative follow-up. Exclusion criteria were: multiple vertebral compression fractures, fracture duration more than three weeks, pathological fractures caused by malignant tumors, severe burst vertebral fractures, vertebral fractures with neurological symptoms, and severe blood coagulation disorders.
Manipulation-reduction for the thoracic osteoporotic fracture was performed with hyperextension position of spine. CT scanning was essential for the design of various preoperative parameters including abduction angle (∠ɑ), inclination angle (∠β), entry point of skin (A) and optimal puncture path (AD) (Figure A and B). Point F marked on the skin was the spinous process of fractured vertebra; and point A, as entry point of skin, was decided by AF distance lateral to midline. Point B was the transverse process tip with point C as the base of the pedicle and point D as the front of the vertebra on this puncture path. Distance of BC could enable surgeon to control the puncture depth of bony part, and beyond the limited depth of CD was of high danger.
Patient was placed in prone position with abdominal suspension by soft pillows under chest and ilium. Both arms were put up to head. The angle of C-arm fluoroscopy machine was adjusted to project toward the injured vertebral body, rather than the whole thoracic column for standard AP and lateral view. In this standard AP view, both pedicle shadows of injured vertebral were symmetrical to midline. In this standard lateral view, the end plate of the injured vertebral body presented "single line shadow". The upper and lower margins of the two pedicles were fully overlapped. Ultrasonic probe was prepared after surgical disinfection. Anesthesia needle entered the skin by point A, which has already been made during the preoperative management. Local anesthesia was carried out with the ultrasound guidance by low frequency convex array ultrasound probe from skin layer, subcutaneous, fascia to periosteum of transverse process.
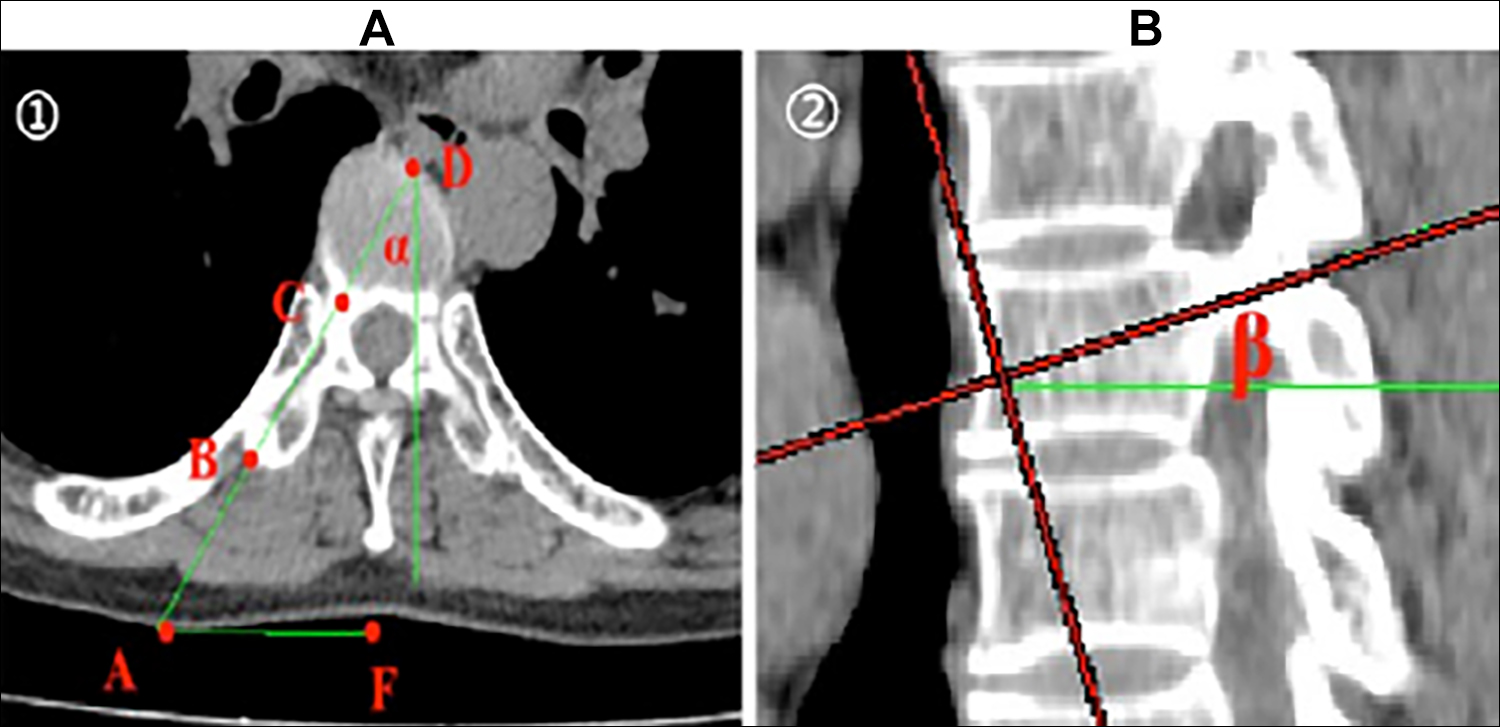 Figure 1: ①. Entry point of skin (A), the tip of the transverse process (B), the base of the pedicle (C), the front of the vertebra on this puncture path (D), mark on the skin of spinous process, abduction angle(<ɑ). Inclination angle (<β).
Figure 1: ①. Entry point of skin (A), the tip of the transverse process (B), the base of the pedicle (C), the front of the vertebra on this puncture path (D), mark on the skin of spinous process, abduction angle(<ɑ). Inclination angle (<β).
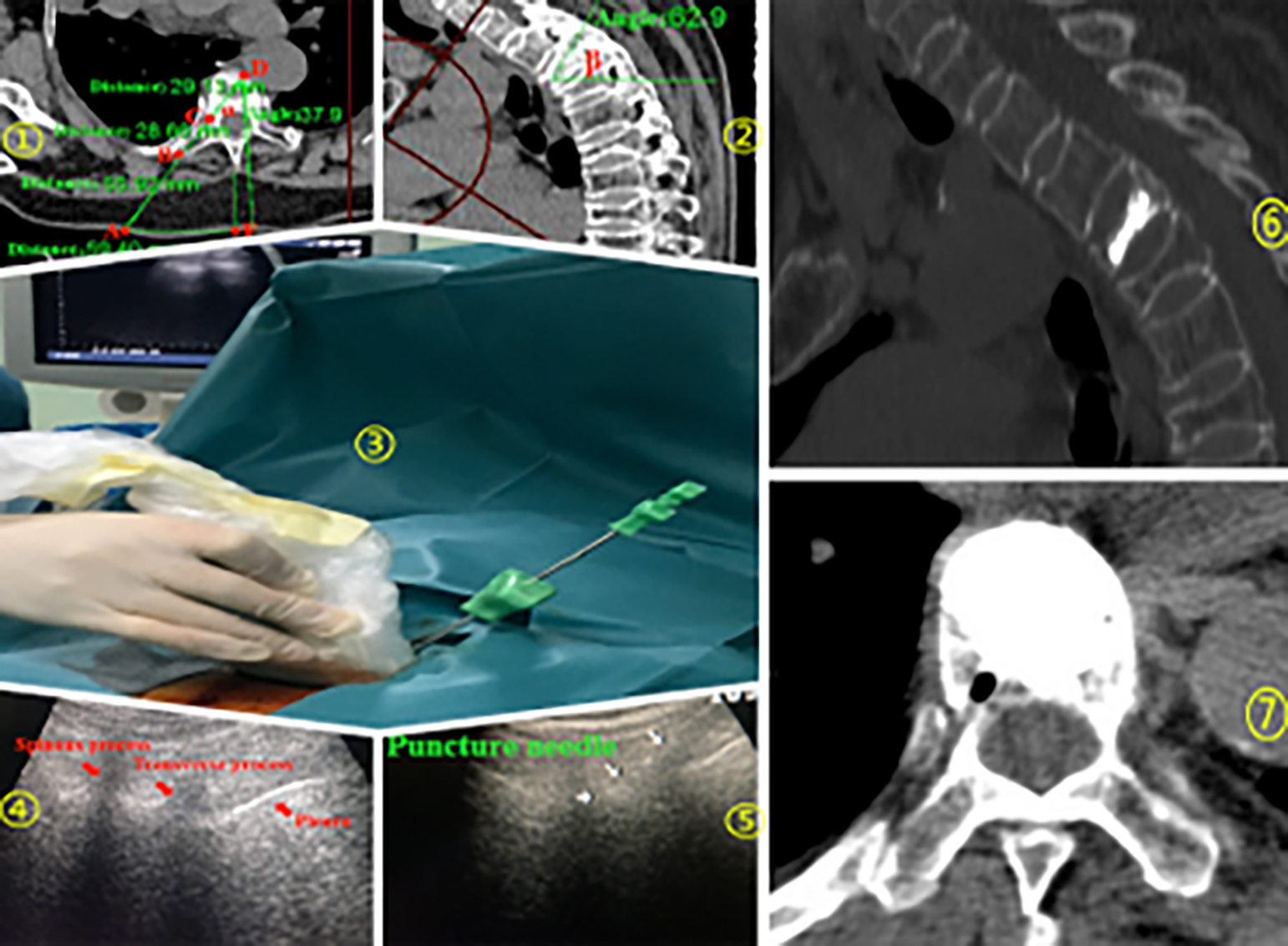 Figure 2: Typical case for T3 fracture, (1,2) Preoperative parameters planning, (3) Ultrasound assistance procedure, (4,5) Ultrasound identification of anatomy and guidance puncture needle, (6,7) Distribution of bone cement.
Figure 2: Typical case for T3 fracture, (1,2) Preoperative parameters planning, (3) Ultrasound assistance procedure, (4,5) Ultrasound identification of anatomy and guidance puncture needle, (6,7) Distribution of bone cement.
Skin was cut for 0.5 mm, and the thin thoracic puncture needle was guided by ultrasound from skin point A through subcutaneous, fascial, muscular layer to precisely reach point B, which was the tip of transverse process. Subsequently, the PVP puncture needle was properly hit into the bony part of this injured thoracic vertebra gradually with the planned head inclination and abduction angle before operation. Entrance depth of PVP puncture needle from point B was continuously observed, according to the measured date from surgery planning procedure. Reasonably, the safe bony advance distance was limited in 10 mm beyond BC, and no more than BD. Ultrasound-guided puncture position was detected by fluoroscopy. Quantities of bone cement were controlled and injected into the vertebral body slowly. The distribution of bone cement was confirmed by fluoroscopy. After the injection, the working sleeve and push rod were rotated together to avoid adhesion with bone cement and pulled out until cement became solid. Incision was locally compressed by hand to stop bleeding for 5 mins and sealed by a small elastic band-aid (Figure 2).
Postoperatively, patients kept to bed for 2-6 hours and wore a brace to get off the bed. Pain, induced by fracture, improved obviously, so the patient was encouraged to walk, breath exercise and left hospital in three days after taking X-ray and CT. Besides the instruction to fall prevention, the systematic anti-osteoporosis treatment should be given to each patient. There were eight patients (6 females, 2 males) with upper or mid-thoracic vertebral body OVCFs (1 each T3 and T6, 2 for T7, and 4 for T8) underwent ultrasound-assisted PVP and finished the follow-up for more than 12 (14.63 ± 0.80) months. The duration of surgery was 23.88 ± 1.67 minutes. There was one case of bone cement leakage, but without neurological symptom. There was no re-fracture of operative and adjacent vertebral body during the follow-up.
DISCUSSION
A common and serious complication of osteoporosis (OP), the OVCF, always induces pain and other complications which may influence patients' life quality or even threaten their lives.6 Although thoracolumbar spine are affected OVCF with high frequency, the incidence rate and risk of thoracic fractures in the upper and middle parts should also not be underestimated.7 Patients with OVCF, who have underwent surgery, reveal 22% lower mortality rate in the next decade as compared with the patients who have been treated by conservative way.8 It has been widely accepted that cementation of vertebral body of PVP is a classical method in treating OVCF due to its less invasiveness and effective pain control. However, owing to particularity of anatomical factors which obviously block the AP and lateral fluoroscopy view, precise puncture of PVP in treating the upper and mid-thoracic OVCF is still a challenge for surgeons.
Scholars have defined T1-T4 as the upper thoracic vertebra, T5-T8 as mid-thoracic vertebra, and T9-T12 as the lower thoracic vertebra.9 Research had shown that unilateral approach was characterised by short operation time, low hospital cost, less number of X-ray views, and less risk of bone cement leakage.10 In this study, unilateral parapedicular approach was adopted. Parapedicular pathway may improve the bone cement distribution in the injured vertebral body, which could reduce the risk of re-fracture of operative and adjacent vertebral body, according to the result of this study.
Puncture has a vital role in the whole PVP procedure. There are many challenges in the process of puncture in treating an upper or mid-thoracic vertebral body injury. The anatomical factors mentioned above are of the first concern. Shapes vary among different segments in thoracic vertebra, and the pedicle of upper and mid-thoracic vertebrae are thinner as compared with lower parts of spine. In addition, the wall of thoracic pedicle is close to spinal cord medially and pleura laterally. There are vital blood vessels, pleura and other important organs in front of vertebral body. In the standard lateral fluoroscopy, the occlusion of scapula and shoulder joint affect the precision of puncture. In the AP view, the overlaping shadow of the sternum, and calcification of aortic arch, which is a common disorder among the elderly, also significantly interferes with the image recognition degree of the injured vertebra. At the same time, osteoporotic vertebral body itself decreases the degree of identification of anatomical structure. Therefore, anatomical factors interfere with identification under the fluoroscopy. Secondly, PVP under fluoroscopy only involves radiation exposure. In addition, fluoroscopy machine moving in and out adds to operation time. Thirdly, fluoroscopy can only produce two-dimensional images, so routine PVP is performed depending on the subjective experience of the surgeon to a large extent. Fourth, there is a posterior medial branch of the spinal nerve at the tip of the transverse process of the thoracic vertebrae. Inaccurate repeated puncture may increase the risk of injury of this nerve and cause postoperative shoulder and back pain. Therefore, it is difficult and risky to treat upper and mid-thoracic vertebral fracture with PVP under fluoroscopy guidance only.
Based on the anatomical and pathological factors, PVP with fluoroscopy alone is still accompanied with several risks in treating upper and mid-thoracic fracture. However, intraoperative ultrasound guidance can make up for many of these deficiencies. Nerve, tendon, ligament, and bone are particularly suitable for evaluation by this technique.11 Portable ultrasonic probe is small in size and can be moved on the operating table easily, which could avoid frequent moving in and out of the large C-arm machine. Meanwhile, conventional soft tissue, overlapping bone (scapula of sternum) and lung texture have always affect fluoroscopy view and also induce the high radiation exposure at the same time. Images generated by ultrasound can avoid these interference factors and provide more intuitive pictures to enable surgeon to identify the bone puncture point. In addition, without large equipment like intraoperative CT or MRI scanning assistance, ultrasound can achieve real-time imaging to dynamically guide accurate anesthesia and PVP puncture, which decreases iatrogenic injury risk and save operation time.12 It has been reported that ultrasound could even check the integrity of the pedicle screw canal.13 Theoretically, it may also detect the bone cement leakage in time. Application of ultrasound assistance in PVP can obviously reduce the frequency of fluoroscopy during the puncture procedure, which can both save the duration of surgery with (23.88±1.67) mins in this study, and radiation exposure. The most important advantage is that the real-time visualisation operation can ensure safety, and avoid iatrogenic injury. Therefore, It is a convenient and safe operation, and it is also conducive to shorten the technique learning curve of PVP for beginners.
This technical operation has the following notices: Patients are under the local anesthesia. In order to obtain well cooperation and guarantee the safety during this procedure, it is essential to perform preoperative introduction and intraoperative communication with patients. Wide shape of ultrasonic probe should be avoided for patients who are very thin, otherwise the ultrasonic probe cannot closely contact the skin on the operation area, which would influence the inaccuracy of imaging. The fracture time and the compressed degree of vertebral body should be limited in this technique, or the effect of the reduction by manipulation would be unsatisfied and add the cement leakage risk as well. The diameter of the puncture needle should be controlled, usage of larger diameter may enhance iatrogenic injury in thoracic vertebra body. One segment upper and lower the fracture vertebra’s pedicles shape in AP view, is helpful in identification of puncture needle position on injury thoracic vertebral body when fluoroscopy, in some serious osteoporosis cases, is used to check the puncture position after ultrasound assistance. Cement leakage is a common complication of PVP, a proper amount of gelatin sponge placed by working sleeve before the injection could reduce the leakage rate. Control bone cement status is vital element in leakage precaution. After the injection, rotate the push rod and the working sleeve and leave them for a while to avoid the leakage of the bone cement from the track. Emphasis on cough control and fall prevention are reasonable precaution measures for avoiding recurrence of thoracic OVCF. Furthermore, systematic effective anti-osteoporosis treatment for these patients is essential as well.
To sum up, intraoperative ultrasound-assisted unilateral parapedicular approach PVP is a feasible method and of obvious advantages in upper and mid-thoracic OVCF. With the assistance of ultrasound, the preoperative design, including skin entry point, abduction angle (∠ɑ), inclination angle (∠β), and depth of puncture, can not only reduce the operational risk, but also shorten the operation duration and dosage of radiation exposure. Additionally, the price of ultrasound equipment is affordable. With the optimisation of ultrasound equipment, this technology not only can guide PVP puncture by dynamic visualisation, but also has a certain prospect in monitoring the leakage of bone cement in time. Therefore, this technique is worthy of clinical promotion for upper and mid-thoracic vertebra body osteoporotic fracture.
DECLARATION:
The application of this technique was approved by the Research Ethics Committee of Hospital (No. 672HREC20181507). The authors certify that they have obtained all appropriate patients’ consent forms. The patient understands that their names and initials will not be published and efforts will be made to conceal their identity. Qilin Lu and Long Chen contributed equally in this study.
CONFLICT OF INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
DZ, LC, QL: Checked the literature and designed the technique.
LC, QL: Wrote the manuscript and revised it.
YZ, XW: Collected the patients’ files and carried out evaluation.
ACKNOWLEDGEMENT:
The authors would like to thank Prof. Xiaojuan Wang for performing this technical operation to all patients.
REFERENCES
- Hoyt D, Urits I, Orhurhu V, Orhurhu MS, Callan J, Powell J, et al. Current concepts in the management of vertebral compression fractures. Curr Pain Headache Rep 2020; 24(5):16. doi: 10.1007/s11916-020-00849-9.
- Saracen A, Kotwica Z. Vertebroplasty (PVP) is effective in the treatment of painful vertebral hemangiomas. Acta Orthop Belg 2018; 84(1):105-7.
- Wang F, Wang LF, Miao DC, Dong Z, Shen Y. Which one is more effective for the treatment of very severe osteo-porotic vertebral compression fractures: PVP or PKP? J Pain Res 2018; 11:2625-31.doi: 10.2147/JPR.S179022.
- Arnold MJ, Jonas CE, Carter RE. Point-of-care ultrasono-graphy. Am Fam Physician 2020; 101:275-85.
- Tiouririne M, Dixon AJ, Mauldin FW, Scalzo D, Krishnaraj A. Imaging performance of a handheld ultrasound system with real-time computer-aided detection of lumbar spine anatomy: A feasibility study. Invest Radiol 2017; 52(8): 447-55. doi: 10.1097/RLI.0000000000000361.
- Sanli I, Kuijk SMJ, Bie RA, Rhijn LW, Willems PC. Percutaneous cement augmentation in the treatment of osteoporotic vertebral fractures in the elderly: A systematic review. Eur Spine J 2020; 29(7): 1553-72. doi: 10.1007/ s00586-020-06391-x.
- Seong JY, Kim JS, Jung B, Lee SH, Kang HY. CT-guided percutaneous vertebroplasty in the treatment of an upper thoracic compression fracture. Korean J Radiol 2009; 10(2):185-9. doi: 10.3348/kjr.2009.10.2.185.
- Hinde K, Maingard J, Hirsch JA, Phan K, Asadi H, Chandra RV. Mortality outcomes of vertebral augmentation (vertebroplasty and/or balloon kyphoplasty) for osteoporotic vertebral compression fractures: A systematic review and meta-analysis. Radiology 2020; 295(1):96-103. doi: 10.1148/radiol.2020191294.
- Liu J, Tang J, Zhang J, Gu ZC, Yu SH. Percutaneous vertebral augmentation for osteoporotic vertebralcompression fracture in the mid-thoracic vertebrae (T5-8): A ret retrospective study of 101 patients with 111 fractured segments. World Neurosurg 2019; 122:1381-7.
- Tang J, Guo WC, Hu JF, Yu L. Unilateral and bilateral percutaneous kyphoplasty for thoracolumbar osteoporotic compression fractures. J Coll Physicians Surg Pak 2019; 29(10):946-50. doi:10.29271/jcpsp.2019.10.946.
- Sahlani L, Thompson L, Vira A, Panchal AR. Bedside ultrasound procedures: Musculoskeletal and non-musculoskeletal. Eur J Trauma Emerg Surg 2016, 42(2): 127-38. doi: 10.1007/s00068-015-0539-3.
- Simard R. Ultrasound imaging of orthopedic injuries. Emerg Med Clin N Am 2020; 38(1):243-65. doi: 10.1016/j.emc. 2019.09.009.
- Kantelhardt SR, Bock CH, Larsen J, Bockermann V, Schillinger W, Rohde V, et al. Intraosseous ultrasound in the placement of pedicle screws in the lumbar spine. Spine (Phila pa 1976) 2009; 34(4):400-7. doi: 10.1097/BRS. 0b013e31819286ca.