T-cell Acute Lymphoblastic Leukemia in an Infant: A Case Report and Review of Literature
By Neelum Mansoor1, Naeem Jabbar2Affiliations
doi: 10.29271/jcpsp.2022.08.S174ABSTRACT
A high index of suspicion of acute leukemia in infants presenting with atypical clinical and laboratory features is of paramount importance to avoid the delay in diagnosis, and treatment of this rare, yet extremely aggressive entity. We report a case of a 10 months infant presenting with a one-month history of fever and restlessness along with complete blood count (CBC) findings of bicytopenia, borderline leukocytosis, leucoerythroblastic picture, and reactive lymphocytes. Considering infection, the patient was kept on antibiotics and symptomatic treatment. However, the persistence of findings led to bone marrow aspirate which revealed T-cell acute lymphoblastic leukemia (T-ALL) on flow cytometry.
Infant ALL is almost B cell phenotype and T-ALL is hardly ever reported. Knowledge and recognition of these cases can help to improve awareness regarding consideration of this rare phenotype of leukemia irrespective of clinical presentation and age of the patient.
Key Words: Acute lymphoblastic leukemia, Infant leukemia, Flow cytometry.
INTRODUCTION
Acute lymphoblastic leukemia (ALL) in infants is a rare disease with reported incidence of 2.3-5% of all childhood ALL.1-3 It is an aggressive entity of poor prognosis that typically presents with high white cell count, hepatosplenomegaly, frequent central nervous system (CNS) involvement, and skin infiltration.4 Another distinct trait of infantile ALL is the presence of MLL gene re-arrangement which may be seen in up to 80% of cases.5 Almost all infantile ALL is of B cell phenotype with T-ALL being exceptionally uncommon in this age group.1-4
Here, we report a case of an infant presenting with fever, restlessness, and atypical complete blood count (CBC) findings, that was diagnosed as T-ALL at 10 months of age. This case is described to highlight the occurrence of this extremely rare phenotype in this age group for the awareness of physicians. Moreover, contrary to typical presentation, hyperleukocytosis and extramedullary manifestations were not seen in this case, which delayed the suspicion and definitive workup. Therefore, reporting this case will also help in eliciting a high index of suspicion among physicians.
CASE REPORT
A 10-month infant presented to a secondary care center with a one-month history of fever and restlessness which progressed to sleep deprivation and tachypnea for the last few days. On examination, no lymphadenopathy or hepatosplenomegaly was observed. During admission, baseline workup including CBC, routine biochemistry, blood culture, and chest X-ray were performed. CBC revealed hemoglobin of 6.9 g/dl, total leukocyte count (TLC), 19×109/L, and platelets 65×109/L along with leucoerythroblastic picture and reactive lymphocytes on peripheral smear. Biochemistry results were within reference limits and there was no growth in blood culture. However, a chest X-ray showed mediastinal widening (Figure 1). The patient was kept on antibiotics and symptomatic treatment including antipyretics, nebulisation, and blood transfusions for almost 10-14 days. Due to the persistence of these findings, bone marrow aspirate was performed which revealed T-ALL on flow cytometry (Figure 2) with the following immunophenotype:
Positive: CD45, TdT, CD34, CD 99, CD1a, Intracellular CD3, CD5, CD4
Negative: CD 79a, CD19, Intracellular MPO, CD13, CD33, CD117, CD10, CD20, CD8
Cytogenetics by fluorescent in-situ hybridization (FISH) was also performed for MLL gene rearrangement and it was reported as negative (Figure 3). The patient was referred to our center for further management. Detailed counseling for curative treatment was done and the patient was kept on the list for the insertion of port-a-cath but the family left against the medical advice (LAMA) on the next day of admission for spiritual/alternative medicine and came back after three weeks with a deteriorated clinical condition, hyperleukocytosis (TLC: 436×109/l with 94% blasts) and findings of tumor lysis syndrome. The patient collapsed within a few hours of presentation and could not be revived.
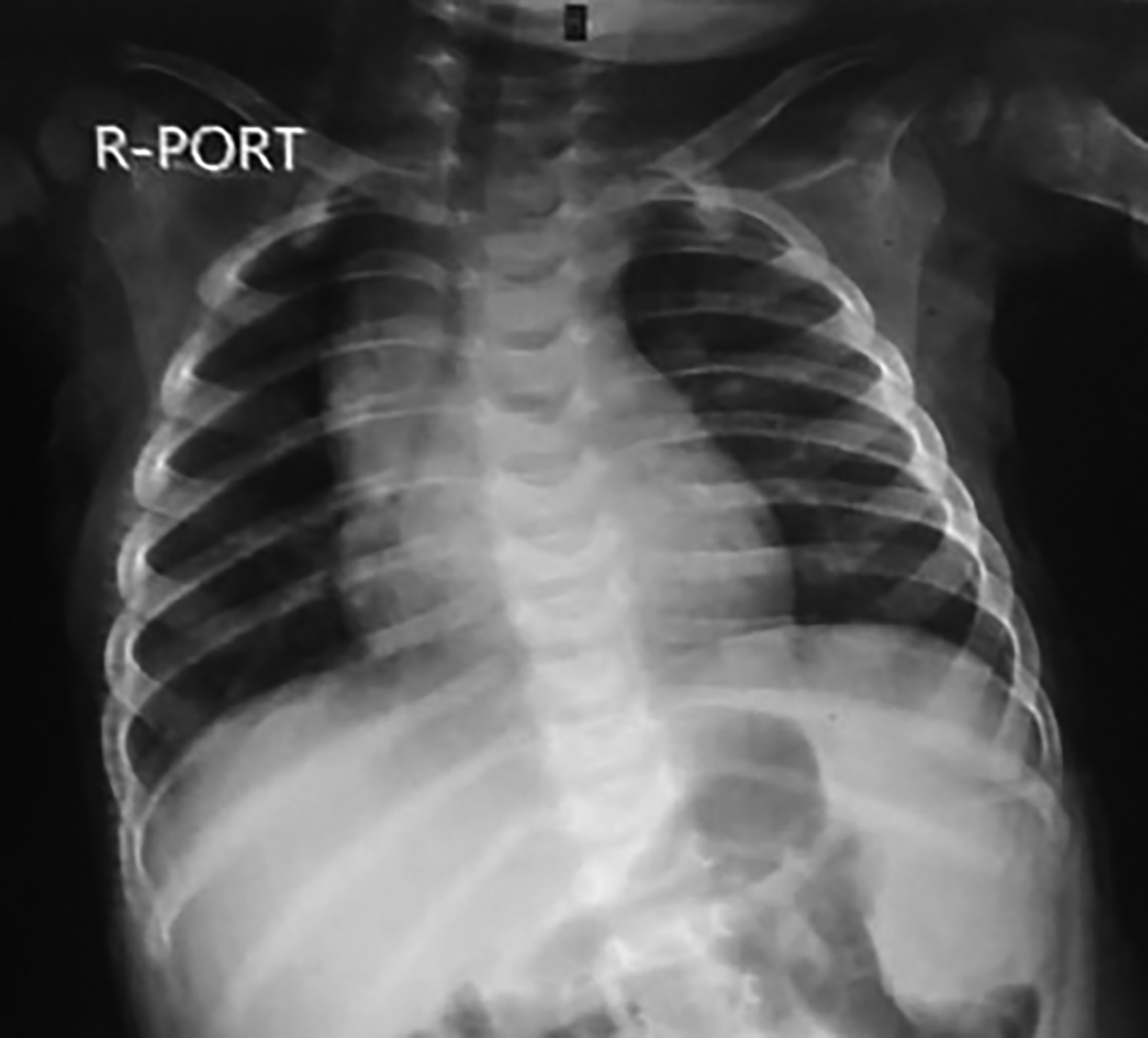 Figure 1: Chest x-ray showing mediastinal widening.
Figure 1: Chest x-ray showing mediastinal widening.
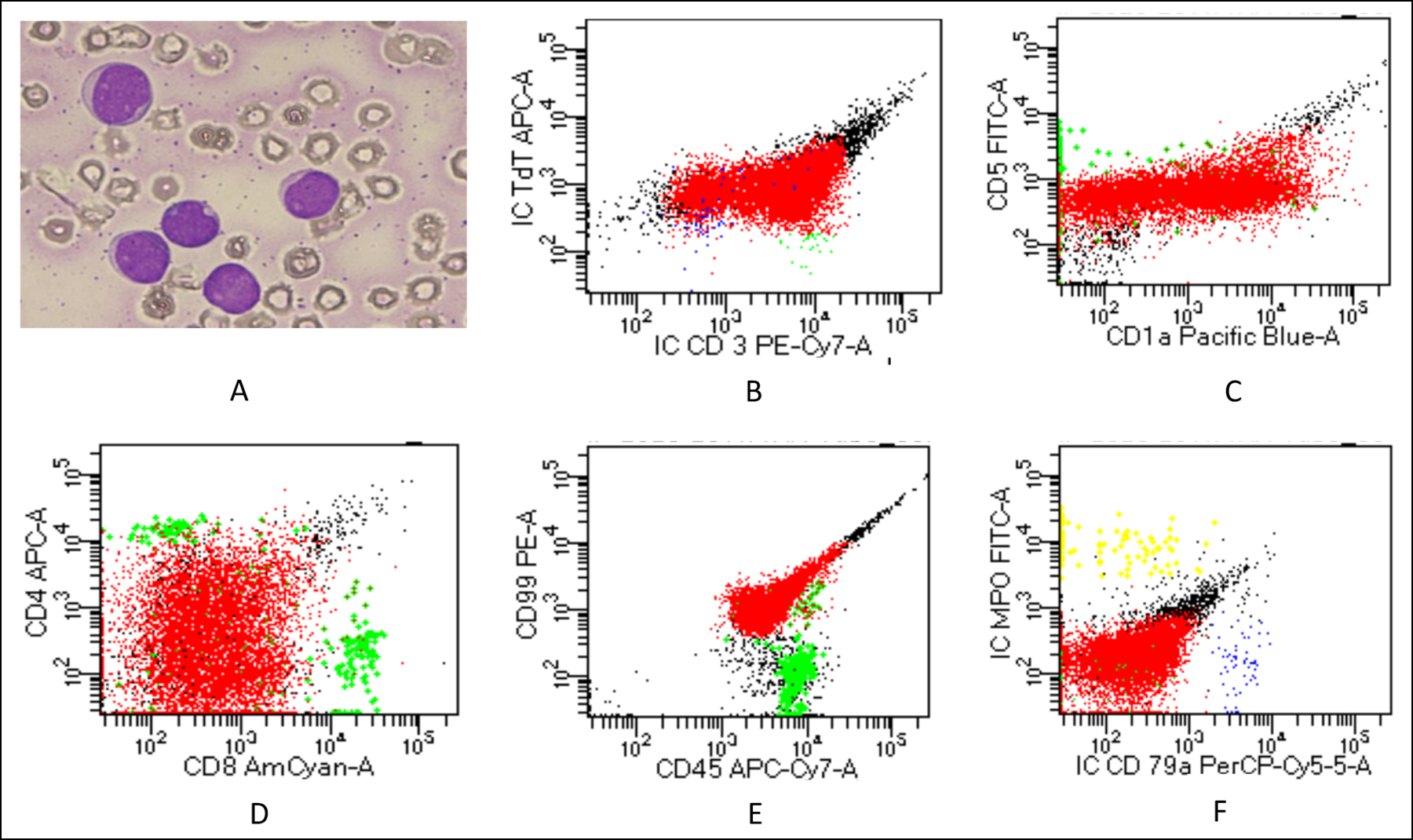 Figure 2: (A) Peripheral smear showing medium sized blasts (B-F) Dot plots of flow cytometry showing cell populations as: Red=Blasts, Green=Benign T lymphocytes, Blue=Benign B lymphocytes, Yellow=Granulocytes. Blasts are TdT +, IC CD3 +, CD5 Dim +, CD1a partial +, CD4 partial +, CD8 -, CD99 +, CD45 +, IC MPO - and ICCD79a -.
Figure 2: (A) Peripheral smear showing medium sized blasts (B-F) Dot plots of flow cytometry showing cell populations as: Red=Blasts, Green=Benign T lymphocytes, Blue=Benign B lymphocytes, Yellow=Granulocytes. Blasts are TdT +, IC CD3 +, CD5 Dim +, CD1a partial +, CD4 partial +, CD8 -, CD99 +, CD45 +, IC MPO - and ICCD79a -.
DISCUSSION
Diverse clinical presentations of ALL have historically constituted an enormous challenge in terms of diagnostic approach as these atypical presentations may be misleading in some cases as observed in our case. Our patient presented with fever and restlessness, which later progressed to tachypnea. The typical clinical findings of infantile ALL including lymphadenopathy, organomegaly, CNS and skin involvement, and hyperleukocytosis were not seen in this case.4
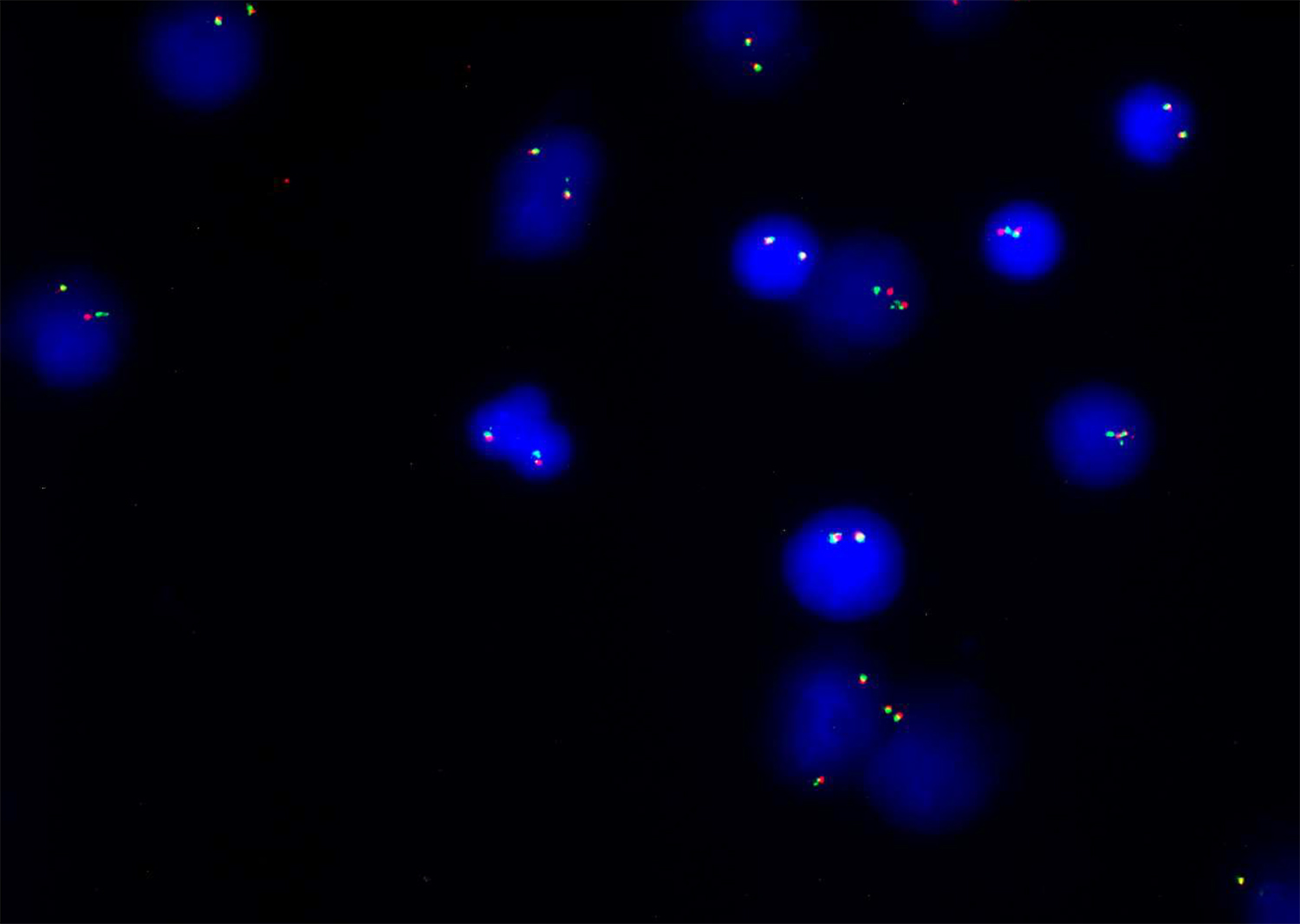 Figure 3: Fluorescence in-situ hybridization image showing normal (2 yellow) fusion signals; MLL (11q23) break apart dual-color gene rearrangement is not detected in 324 nuclei examined.
Figure 3: Fluorescence in-situ hybridization image showing normal (2 yellow) fusion signals; MLL (11q23) break apart dual-color gene rearrangement is not detected in 324 nuclei examined.
Infantile leukemia is, fortunately, a rare disease with a slight predominance of lymphoid over myeloid subtype. Among the reported lymphoid cases, nearly all are of B cell phenotype with less than 5% of T-ALL, an entity commonly seen in older children and adolescents.1-4 Interestingly, infantile leukemia demonstrates a relatively higher prevalence in females in contrast to male preponderance that is observed beyond the first year of life.6
The immunophenotypic analysis in our case showed positivity for immature markers TdT, CD34, CD99, and CD1a and T lineage markers including CD3, CD5, and CD4. Like the reported literature, our patient did not show CD10 expression.7 In cytogenetic analysis, the presence of MLL gene rearrangement (also called KMT2A) is a consistent cytogenetic finding irrespective of subtype of infantile leukemia (lymphoid/myeloid); however, contrary to reported literature our patient did not show this cytogenetic abnormality.4,7
There is a remarkable variation in the clinical outcome of ALL in terms of age at diagnosis.8 Historically, results of large studies showed dismal outcomes for infantile ALL as compared to older children. The vulnerability of these young children to complications and high rate of toxicity, and relapse are the major challenges.3,8 Current treatment protocols for infantile ALL are quite different and toxic from general childhood ALL protocols. There are three major groups conducting clinical trials for infantile ALL and all have adopted an identical induction therapy based on interfant-99.9 In recently completed trials, a prospective risk-stratified approach was used that incorporates MLL gene rearrangement status and age.4 The use of hematopoietic stem cell transplantation (HSCT) in infantile leukemia showed uncertainty regarding the risk versus benefit ratio and there is limited evidence of better outcomes with HSCT in a small sub-group of patients that have a high risk of relapse.10 Immunotherapy, targeting the RAS pathway, and the use of FLT3 inhibitors are some of the novel treatment approaches under investigation to improve the standard of care in infantile ALL.11
ALL, in infants, is almost B cell phenotype with aggressive clinical behavior and poor outcome. Due to the extremely rare occurrences, little is known about T-ALL in infants. Thus, more multi-centre studies and case series are needed to have better insights of this phenotype in terms of disease biology, clinical course, and survival outcome. Moreover, atypical presentation can be a feature of infantile ALL which demands a high index of suspicion for early diagnosis and treatment initiation.
PATIENT’S CONSENT:
Informed consent was obtained from the father to publish this case report.
COMPETING INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
NM: Concept, manuscript writing, and proofreading.
NJ: Data collection and proof reading.
Both authors approved the final version of the manuscript to be published.
REFERENCES
- Cherungonath A, Appaji L, Padma M, Arunakumari BS, Arunkumar AR, Avinash T, et al. Profile of acute lympho-blastic leukemia in children under 2 years of age. Indian J Med Paediatr Oncol 2018; 39:307-11.
- Fadoo Z, Nisar I, Yousuf F, Lakhani LS, Ashraf S, Imam U, et al. Clinical features and induction outcome of childhood acute lymphoblastic leukemia in a lower/middle income population: A multi-institutional report from Pakistan. Pediatr Blood Cancer 2015; 62(10):1700-8. doi: 10.1002/ pbc.25583.
- Brethon B, Cave H, Fahd M, Baruchel A. Infant acute leukemia. Bull Cancer 2016; 103(3):299-311. doi: 10.1016/j.bulcan.2015.11.009.
- Brown P, Pieters R, Biondi A. How I treat infant leukemia. Blood 2019; 133(3):205-14. doi: 10.1182/blood-2018-04- 785980.
- Britten O, Ragusa D, Tosi S, Kamel YM. MLL-rearranged acute leukemia with t (4; 11) (q21; q23) — Current treatment options. Is there a role for CAR-T cell therapy? Cells 2019; 8(11):1341. doi: 10.3390/cells8111341.
- Howlader N, Noone AM, Krapcho M, Miller D, Bishop K, Kosary CL, et al. SEER cancer statistics review, 1975-2014, National Cancer Institute. Bethesda, MD, https://seer.cancer. gov/csr/1975_2014/, based on November 2016 SEER data submission, posted to the SEER web site, April 2017.
- Vijayanarasimha D, Madhumathi DS, Kumari P, Naik J, Appaji L, Lakshmidevi V. The pheno-genotypic characteristics of infantile acute leukemia in a regional cancer center from South India. J Appl Hematol 2017; 8:61-7.
- Hossain MJ, Xie L, Mc Cahan SM. Characterisation of pediatric acute lymphoblastic leukemia survival patterns by age at diagnosis. J Cancer Epidemiol 2014; 2014:865979. doi: 10.1155/2014/865979.
- Pieters R, Schrappe M, De Lorenzo P, Hann I, De Rossi G, Felice M, et al. A treatment protocol for infants younger than 1 year with acute lymphoblastic leukaemia (Interfant-99): An observational study and a multicentre randomised trial. Lancet 2007; 370(9583):240-50. doi: 10.1016/S0140-6736(07)61126-X.
- Sison EA, Brown P. Does hematopoietic stem cell transplantation benefit infants with acute leukemia? Hematology Am Soc Hematol Educ Program 2013; 2013:601-4. doi: 10.1182/asheducation-2013.1.601.
- Driessen EM, van Roon EH, Spijkers- Hagelstein JA, Schneider P, de Lorenzo P, Valsecchi MG, et al. Frequencies and prognostic impact of RAS mutations in MLL rearranged acute lymphoblastic leukemia in infants. Haematologica 2013; 98(6):937-44. doi: 10.3324/haematol.2012.067983.