Subglottic Hemangioma: Now You See it, Now You Don’t
By Mawaddah AzmanAffiliations
doi: 10.29271/jcpsp.2022.04.67ABSTRACT
Subglottic hemangiomas may be missed on flexible laryngoscopy or even bronchoscopy requiring a contrast-enhanced computed tomography scan to clinch the diagnosis. We report an otherwise healthy three-month baby girl presenting with recurrent upper airway obstruction with a history of multiple admissions requiring intubation and artificial ventilation. An initial contrast-enhanced computed tomography scan was performed when the child was intubated, which showed no airway anomalies. Subsequent flexible laryngoscopy and diagnostic bronchoscopy showed features of laryngomalacia and subglottic edema; hence, an endoscopic laser-assisted supraglottoplasty was performed. Following the surgical intervention, the child had persistent stridor and a repeat contrast-enhanced computed tomography scan clinched a diagnosis of a localised subglottic hemangioma at the age of 7 months. In the presence of an endotracheal tube, airway hemangiomas may be missed on bronchoscopy and contrast-enhanced computed tomography scan.
Key Word: Subglottic stenosis, Congenital, Hemangiomas, Computed tomography.
INTRODUCTION
Airway hemangiomas are an important cause of upper airway obstruction presenting in the neonatal period.1 Although diagnostic bronchoscopy may show a typical feature of a subglottic mass with a purple, blue or pinkish hue, atypical feature can be seen,1,2 especially when the airway has been tempered by the presence of an endotracheal tube or prior instrumentations. Contrast-enhanced computed tomography (CECT) is a valuable non-invasive method for the evaluation of airway hemangiomas in patients with equivocal findings on bronchoscopy.3 We present a case demonstrating potential limitations of bronchoscopy and CECT as primary methods in establishing the diagnosis of airway hemangiomas.
CASE REPORT
A three-month baby girl was referred to the otolaryngology team following her second admission for severe croup where she had to be intubated. The child was born full-term with a good Apgar score. The mother first noted noisy breathing shortly following birth; however, the baby was thriving well with good suckling and normal cry. A CECT was performed when the child was intubated (Figure 1a), where no airway anomaly was found.
Prior to allowing extubation, a diagnostic bronchoscopy was performed showing subglottic edema with no other associated anomalies. She was subsequently extubated and a dynamic flexible laryngoscopy concluded the features of laryngomalacia. The baby was sent home with residual mild biphasic stridor.
The child later underwent endoscopic laser-assisted supraglottoplasty at the age of four months due to recurrent admissions with worsening upper airway obstruction confounded by upper respiratory tract infections. Following the surgery, she continued to have biphasic stridor. Finally, she was diagnosed with right subglottic hemangioma at 7 months on flexible laryngoscopy, supported by neck CECT findings (Figure 1b), showing an enhancing mass at the right subglottic wall, causing subsequent narrowing of the subglottic airway. She was subsequently treated with oral propranolol and has shown response towards the treatment, without the need of a tracheostomy.
DISCUSSION
The management of a child presenting with upper airway obstruction is often challenging as it represents a potentially life-threatening event requiring a multi-disciplinary approach.4 Common causes of congenital stridor include laryngeal cysts, atresia and stenosis, vocal fold immobility, and subglottic hemangiomas.5 More than one airway abnormality may coexist in a particular patient with congenital airway anomalies in up to 45.2% of the cases.5 This further complicates the case with higher likelihood of multiple presentations with airway distress.
More often than not, in a case of congenital stridor presenting in distress, securing the airway becomes the primary aim as this represents an airway emergency. At this point, careful consideration and quick discussion amongst paediatrician, neonatologist, anaesthetist and an otolaryngologist are prudent to avoid mishaps and choose the best way to secure the airway.4 In the setting of airway distress, non-invasive ventilation with continuous positive pressure may buy time to alert the necessary personnel for a diagnostic and therapeutic micro-laryngoscopy and bronchoscopy. The advantage of performing a diagnostic scope prior to any instrumentation or intubation includes preservation of endoscopic findings peculiar to the disease at hand, allowing the otolaryngologist a first-hand view of the airway.
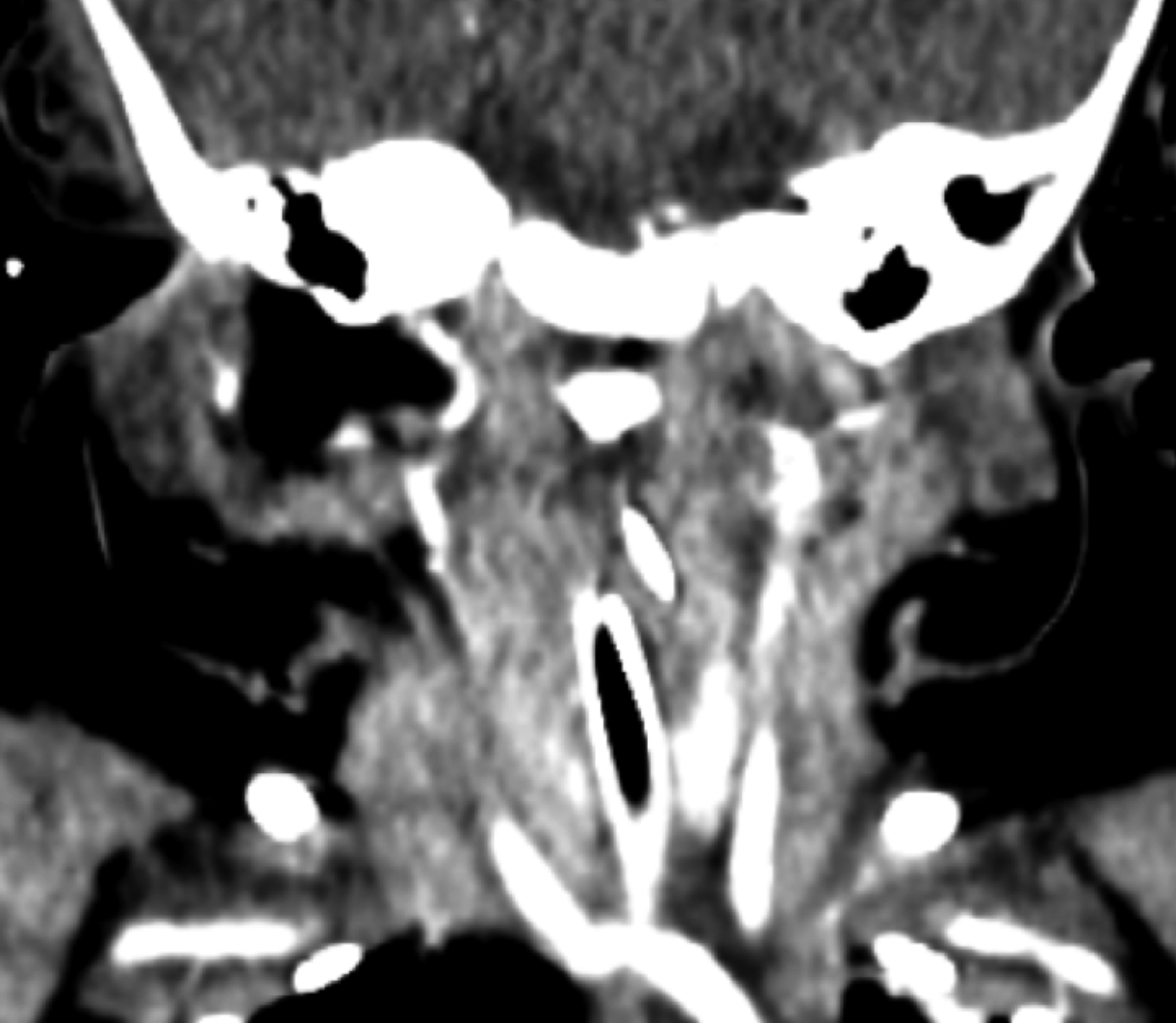 Figure 1a: Coronal-cut contrast-enhanced computed tomography (CECT) neck performed at 3-month in the presence of an endotracheal tube showing no abnormal enhancement suggestive of subglottic hemangioma.
Figure 1a: Coronal-cut contrast-enhanced computed tomography (CECT) neck performed at 3-month in the presence of an endotracheal tube showing no abnormal enhancement suggestive of subglottic hemangioma.
A focal subglottic hemangioma, such as in our case, usually presents with a typical endoscopic finding of a protruded mucosa with purple, blue or pinkish hue. Owing to the soft consistency of these vascular lesions,6 they are easily compressible. We postulate that in the presence of an endotracheal tube, the hemangioma was compressed and that a diagnosis of subglottic hemangioma was missed during the initial bronchoscopy and CECT. Additionally, the loose areolar tissue of the subglottic airway present in a neonate makes it more susceptible to airway edema following intubation.5 We feel that the airway edema may have obscured the finding of a subglottic hemangioma in the initial bronchoscopy. Another reason for the evolving changes seen in the endoscopic and radiologic findings in this patient is the natural history of airway infantile hemangioma itself. The typical presentation of a subglottic hemangioma is of a child with recurrent “croup” presenting with biphasic stridor at 4 to 8 weeks of age. These lesions are characterised by a proliferative phase followed by spontaneous involution.4 The proliferative phase is associated with hypercellularity and increased endothelial turnover with mast cell proliferation in the basement membrane.2 As such, it is during the proliferative phase that these lesions represent a clinically apparent growth, which may have yet to start during the initial bronchoscopy and CECT, which was performed at the age of three months.
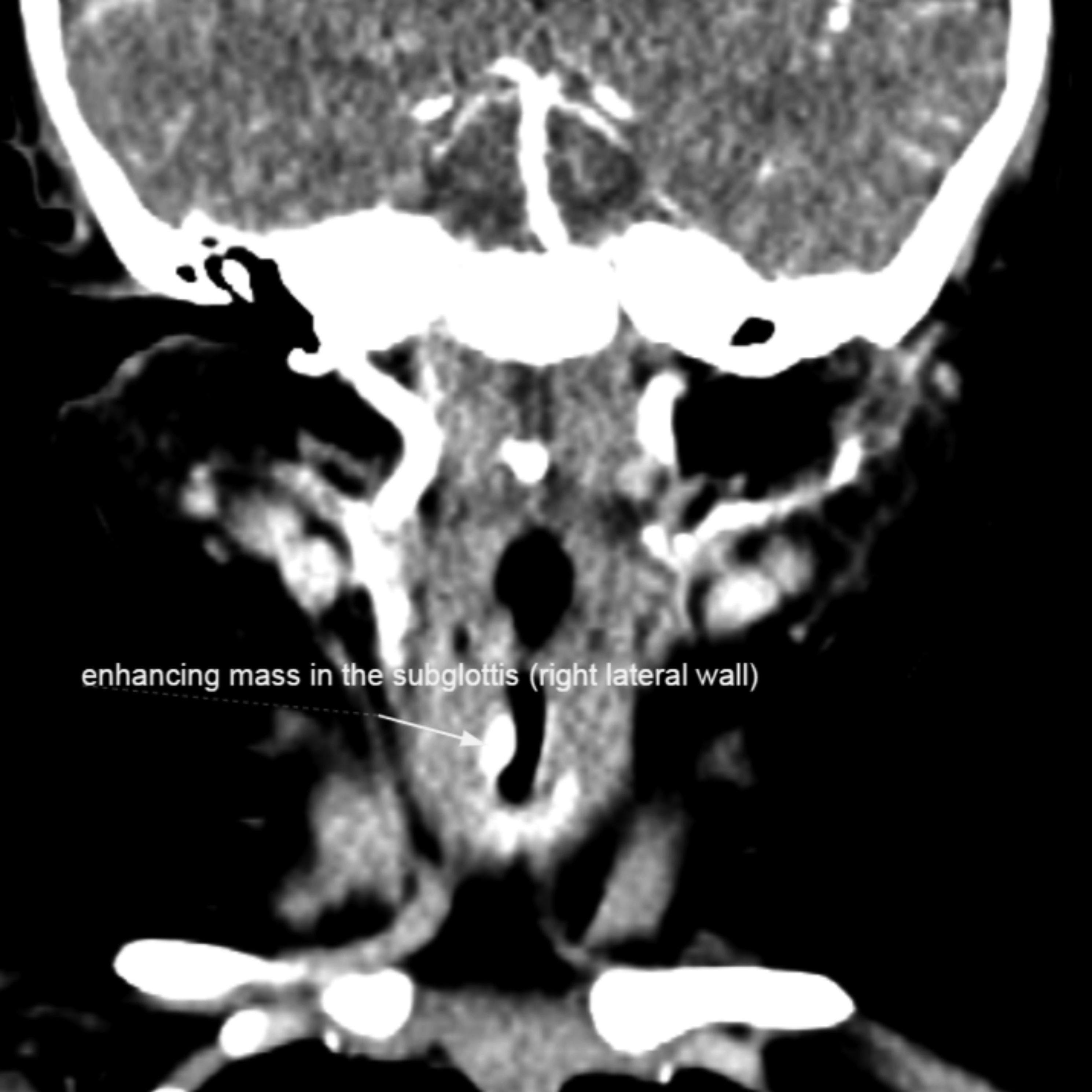 Figure 1b: Coronal-cut contrast-enhanced computed tomography (CECT) performed on the same patient at 7-month showing presence of an enhancing mass at the right lateral subglottic wall with compression of the subglottic airway. Comparing with figure 1a, this enhancing mass might have disappeared in the presence of an endotracheal tube.
Figure 1b: Coronal-cut contrast-enhanced computed tomography (CECT) performed on the same patient at 7-month showing presence of an enhancing mass at the right lateral subglottic wall with compression of the subglottic airway. Comparing with figure 1a, this enhancing mass might have disappeared in the presence of an endotracheal tube.
The management of subglottic hemangiomas has significantly evolved in the last 10 years where dramatic response to propranolol has been shown to reduce the need of tracheostomy in these children.4 In general, only 10% of children with subglottic hemangiomas require surgical intervention, which may include carbon dioxide, potassium-titanyl-phosphate laser excision and intra-lesional steroid or sclerosant injection.2 When treating patients with subglottic hemangiomas, less morbid interventions are deployed, as spontaneous involution is the expected norm.2,4
PATIENT’S CONSENT:
Informed consent from the legal guardian of the patient was obtained to publish the data concerning this case.
CONFLICT OF INTEREST:
The author declared no conflict of interest.
AUTHOR’S CONTRIBUTION:
MA: Managed the case, produced, edited and approved the manuscript in its final state.
REFERENCES
- Wiatrak BJ. Congenital anomalies of the larynx and trachea. Otolaryngol Clin North Am 2000; 33(1):91-110. doi: 10.1016/s0030-6665(05)70209-7.
- Sie KCY, Tampakopoulou DA. Hemangiomas and vascular malformations of the airway. Otolaryngol Clin North Am 2000; 33(1):209-20. doi: 10.1016/s0030-6665(05)70216-4.
- Benjamin ZK , Chaim S, Benjamin SS, Avraham A, Kammal U, Elie P, et al. CT of hemangiomas of the upper airways in children. Am J Roentgenol 2005; 184(2):663-70. doi: 10.2214/ ajr.184.2.01840663.
- Matija D, Alan C. Neonatal stridor. Int J Pediatric 2011; 2012.doi: 10.1155/2012/859104.
- Holinger LD. Etiology of stridor in the neonate, infant and child. Ann Otol Rhinol Laryngol 1980; 89(5):397-400. doi: 10.1177/000348948008900502.
- Richter GT, Friedman AB. Hemangiomas and vascular malformations: Current theory and management. Int J Pediatr 2012; 2012:645678. doi: 10.1155/2012/645678.