Persistent Left Superior Vena Cava: A Rare Finding for the Internists
By Syed Mohammad HaleemAffiliations
doi: 10.29271/jcpsp.2022.08.S104ABSTRACT
A case of a South Asian quinquagenarian gentleman is reported, who was admitted with septic shock and displayed a TYPE II persistent left superior vena cava (PLSVC) after insertion of a central venous catheter, diagnosed on the basis of chest X-ray findings and dilated coronary sinus on transthoracic echocardiography. As the patient was in septic shock, the central venous catheter was retained and vasopressor therapy was instituted with careful hemodynamic monitoring. No immediate or delayed complications attributable to the catheter were observed. Treating physicians should be aware of a possible PLSVC, particularly when a guide wire or a catheter takes a left parasternal course.
Key Words: Persistent left superior vena cava, Septic shock, Central venous catheter.
INTRODUCTION
Persistent left superior vena cava (PLSVC) is an uncommon vascular anomaly.1 It has the potential to complicate central venous catheter and pacemaker insertions but usually remains asymptomatic if undetected.2,3 It occurs as a result of unsuccessful closure of the left superior cardinal vein caudal to the innominate vein. It can be seen as a single defect or in association with atrial septal defect (ASD), bicuspid aortic valve, and coarctation of the aorta. Other defects associated with it include coronary sinus ostial atresia and cor triatriatum.4,5 Confirmation of the diagnosis usually requires saline contrast echocardiography or CT venography. One of the differentials for a dilated coronary sinus on echocardiography is PLSVC. We, herein, report a case in which this condition was encountered in a patient who presented with septic shock after placement of a central venous catheter via the left internal jugular vein. The patient had a dialysis catheter in the right internal jugular vein, so when central venous pressure (CVP) line was required; it was inserted in the left internal jugular vein. On X-ray, it followed a different course than usual, on-call doctor removed it and again passed it. Later it was found out that the patient had PLSVC. It is important for the residents and trainee physicians to know this anomaly when accessing the left internal jugular vein to avoid any confusion.
CASE REPORT
A 62-year Asian gentleman presented to the emergency department with the complaints of dyspnea, generalised body swelling, and fever for two days. His past medical history was significant for uncontrolled diabetes mellitus type 2 (T2DM) for 15 years, hypertension, chronic coronary syndrome, peripheral arterial disease, and chronic kidney disease (CKD) IV (eGFR 25 ml/min/1.73m2). He underwent a coronary angiogram 04 years back for an acute coronary syndrome and he was managed with drug-eluting stents to the right coronary artery and left anterior descending artery. Two weeks before his current hospitalisation, he was commenced on regular hemodialysis via a double lumen catheter placed in the right femoral vein for worsening CKD and anuria. On examination, the patient had altered mental status, a heart rate of 116 beats per minute, blood pressure of 100/60 mmHg, respiratory rate of 32 breaths per minute, and a temperature of 37.5°C. His oxygen saturation was 93% on 10 L of supplemental oxygen via face mask. Systemic examination revealed bilateral fine inspiratory crackles on the chest and his right leg was amputated below the knee joint. The peripheral extremities were cool without mottling. As there was high clinical suspicion of central line-associated bloodstream infection leading to sepsis, the right femoral double lumen catheter, which was in place for two weeks, was removed and replaced with another hemodialysis catheter in the right internal jugular vein. The patient was reviewed by the nephrology service and it was planned to dialyse him with ultrafiltration. During hemodialysis, there was a drop in blood pressure to 70/40 mmHg, so dialysis was discontinued. A left internal jugular central venous catheter (CVC) was inserted for CVP measurement and vasopressor therapy. The CVC was inserted with the modified Seldinger technique under ultrasound guidance. Before passing the guide wire, the venous return was confirmed by noticing the dark red non-pulsatile blood return with the introducer needle. After cannulation, brisk dark (venous) blood return was noted on aspiration from the three catheter ports. However, routine post-procedure chest X-ray (Figure 1) showed that the central venous catheter followed a left paramediastinal course from the left side of the neck.
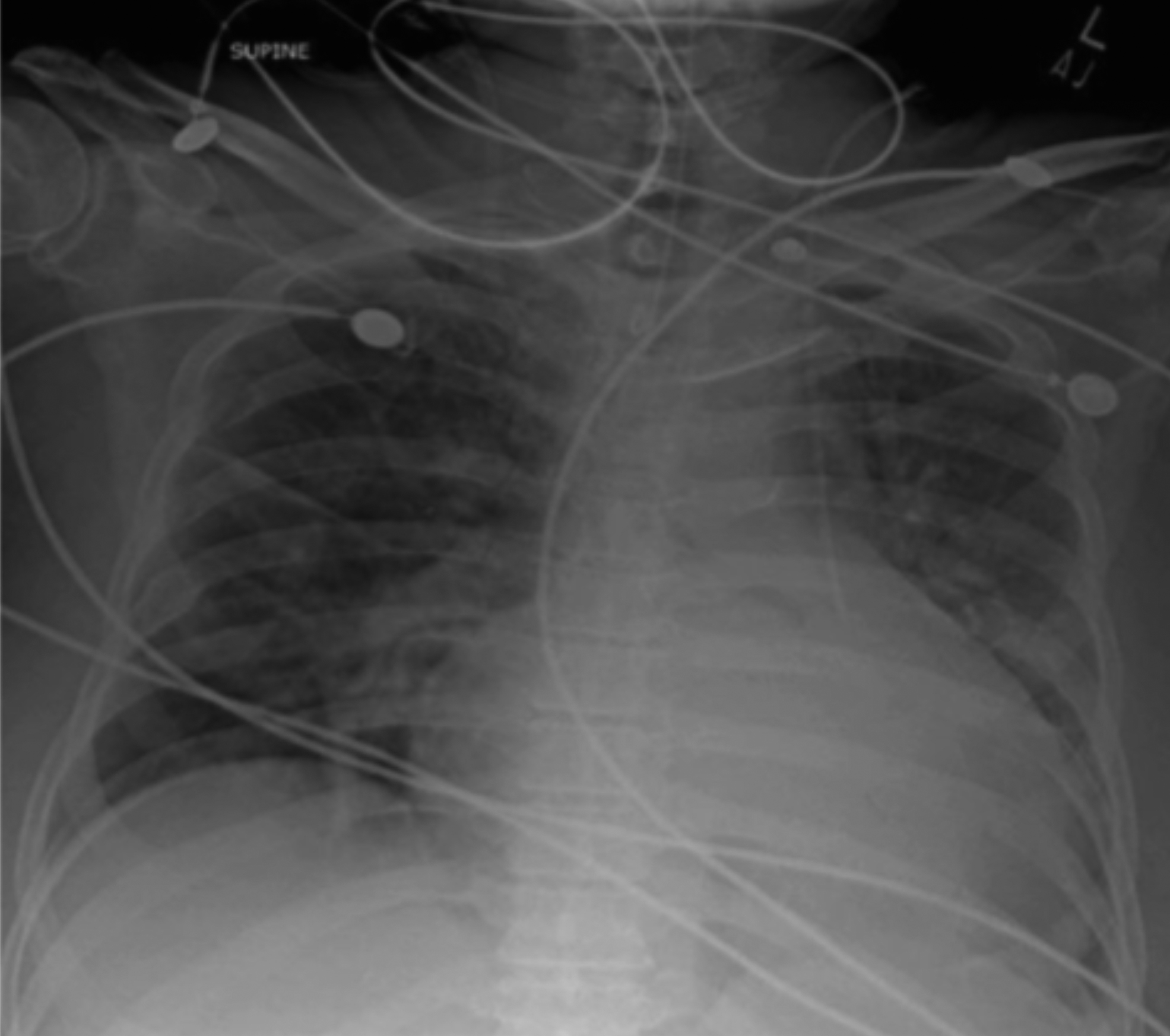 Figure 1: Chest X-ray showing left parasternal course of central venous catheter. In the emergency department, blood gas analysis from CVC showed pH = 7.19; PCO2: 29 mmHg; PO2: 70 mmHg; and CHCO3: 12mmol/l indicative of a venous sample showing severe metabolic acidosis. His blood urea nitrogen (BUN) was 83 mg/dl with a blood creatinine level of 9.8 mg/dl.
Figure 1: Chest X-ray showing left parasternal course of central venous catheter. In the emergency department, blood gas analysis from CVC showed pH = 7.19; PCO2: 29 mmHg; PO2: 70 mmHg; and CHCO3: 12mmol/l indicative of a venous sample showing severe metabolic acidosis. His blood urea nitrogen (BUN) was 83 mg/dl with a blood creatinine level of 9.8 mg/dl.
The CVC was removed and reinserted via the left internal jugular using the same protocol when the shift changed. The chest X-ray remained the same. Meanwhile, vasopressors and inotropes were continued via the CVC. Transthoracic echocardiogram demonstrated a left ventricle with a severely reduced systolic function and an ejection fraction of 20%.
The right ventricular systolic function was reduced (TAPSE 13mm) associated with severe tricuspid regurgitation and pulmonary artery hypertension. In addition, a dilated coronary sinus was identified. To investigate the diagnosis, agitated saline bubbles were injected in the left forearm antecubital vein and the corresponding echocardiogram showed filling of the coronary sinus before the right ventricle which was consistent with the PLSVC. There was no evidence of any other congenital defect. The patient was declared unsuitable for CT venogram in view of overall condition.
Initially, when the chest x-ray revealed that the CVC followed a left paramediastinal course from the left side of the neck, there were following differential diagnoses: (i) Misplaced CVC (ii) left carotid artery catheter cannulation, and (iii) PLSVC.
The patient was electively intubated due to the Glasgow coma scale (GCS) dropping from 12 to 8/15 and transferred to the intensive care unit (ICU) for further management. Plain CT head ruled out cerebrovascular accident (CVA). He was managed with broad-spectrum antibiotics in the renal adjusted doses and remained on vasopressor support of norepinephrine and vasopressin undergoing continuous renal replacement therapy (CRRT) on mechanical ventilation.
Despite aggressive treatment, patient did not improve. The patient developed pulseless electrical activity and expired despite the standard resuscitation efforts.
DISCUSSION
Central venous catherisation is a frequently performed procedure in the management of critically ill patients in medical high care units. PLSVC was discovered in the patient when central venous access was obtained in the left internal jugular vein. It is usual to find it in high-volume cardiac centers on routine insertions of CVCs, pulmonary artery catheters, and pacemakers but not common in high-care medical units.1,2 It is rarely found in isolation while in about 4% it is associated with other congenital heart diseases. Usually, the left SVC drains into the right atrium via the coronary sinus and in 10% of the patients, it drains into the left atrium resulting in partial anomalous systemic venous return. The latter form is usually accompanied by ASD and aortic arch anomalies. Several non-invasive modalities are available to establish the diagnosis of PLSVC. On a plain radiograph, it can be seen a focal widening on the left side of the aortic knob and unusually prominent ascending aorta. A densely opacified and dilated coronary sinus on the contrast-enhanced CT or MRI scan also points towards the diagnosis when contrast is given in the left arm vein. The presence of PLSVC has important clinical implications.3-5 A single-center study on complications of permanent cardiac pacing in patients with PLSVC, underscored the need for a thorough evaluation of the thoracic venous system before intervention in this condition. Although a vast majority of the PLSVCs drain in the right atrium, drainage in the left atrium in the absence of right SVC can lead to the systemic dispersion of emboli bypassing the lungs. Moreover, cardiac dysrhythmias, coronary sinus thrombosis, cardiac tamponade, and fatal cardiac arrest, have been encountered especially when a guidewire or catheter is manipulated via PLSVC.
In conclusion, physicians should recognise the possibility of PLSVC particularly, when a guidewire or a catheter takes a left paramediastinal path on the chest X-ray. Bubble contrast echocardiography should be done to rule out the condition if transthoracic echocardiography shows dilated coronary sinus.
PATIENT’S CONSENT:
Informed consent was obtained before writing the case report.
COMPETING INTEREST:
The author declared no competing interest.
AUTHOR’S CONTRIBUTION:
SMH: Collection of data and writing the case report.
REFERENCES
- Goyal SK, Punnam SR, Verma G, Ruberg FL. Persistent left superior vena cava: A case report and review of literature. Cardiovasc Ultrasound 2008; 6:50. doi: 10.1186/1476- 7120-6-50.
- Asbach S, Biermann J, Giesler U, Baumann T, Zehender M, Bode C, et al. Persistent left superior vena cava: An unusual but conquerable obstacle in device implantation. Clin Res Cardiol 2009; 98(4):268-70. doi: 10.1007/s00392-009- 0755-0.
- Fares WH, Birchard KR, Yankaskas JR. Persistent left superior vena cava identified during central line placement: A case report. Respir Med CME 2011; 4(3):141-3. doi: 10.1016/j.rmedc.2010.11.003.
- Duymus M, Yesilkaya Y, Orman G, Bozkurt A, Yilmaz O. Persistent left superior vena cava draining to the left atrium: A case report and review of the literature. Pol J Radiol 2012; 77(4):65-6. doi: 10.12659/pjr.883632.
- Mudaraddi RR, Greiss HF. A rare case report of unusual path of left internal jugular central line. Indian J Anaesth 2021; 65(2):176-8. doi: 10.4103/ija.IJA_639_20.