Incidence of Silent Ischemic Infarct after Diagnostic Conventional Cerebral Angiogram
By Fawaz Al-Hussain, Bandar Aljafen, Adel Alhazzani, Yousef MohammadAffiliations
doi: 10.29271/jcpsp.2021.03.314ABSTRACT
This study was carried out to determine the risk of new ischemic events, demonstrated by diffusion weighted magnetic resonance imaging (DW-MRI) in patients undergoing diagnostic conventional cerebral angiogram. MEDLINE, EMBASE, Current Contents, Cochrane Central, Expert Files, and bibliographies of included articles were systematically searched to identify prospective studies that reported MRI findings after diagnostic angiogram. Eligibility criteria included the following elements: manuscripts published between 1999 and 2019; prospective design; only diagnostic angiogram imaging performed prior to the MRI; DW-MRI done before and after the angiogram procedure; and number of new diffusion weighted lesions documented after each procedure. Six studies met the eligibility criteria. The total number of diagnostic angiograms reported was 430. DWI lesions were associated with diagnostic angiograms in 106 (24.65%) of total procedures. Mean time fluoroscopy, procedure by resident operator, and vascular risk factors were the recognised independent risk factors for silent cerebral ischemia after diagnostic angiogram.
Key Words: Stroke, Diffusion magnetic resonance imaging, Cerebral infarction, Angiography, Brain ischemia.
INTRODUCTION
Stroke remains one of the five most common non-communicable diseases, as well as one of the leading causes of death worldwide.1 It is also the leading cause of long term disability in adults. As the population is ageing, the overall incidence of stroke is on the rise. Additionally, the progressive improvement in the economy of the most populated countries of the world, China and India, would definitely enhance their access to unhealthy Western diet. It is predicted, therefore, that the stroke incidence in the world will further increase in the coming years.2
Surgical interventions, such as carotid endarterectomy and aneurysm clipping have been for many decades, the sole treatment modality for most cerebrovascular disorders.3,4 However, the last two decades have witnessed the introduction of endovascular procedure (EP), which is much less invasive treatment. This technique has been gradually replacing invasive surgical interventions, and is rapidly becoming the standard of care. The diagnostic conventional angiogram (DCA) has been considered, for many decades, the gold standard technique for evaluation of various cerebrovascular diseases.
However, in the last two decades less invasive and sensitive imaging tools such as CT angiogram (CTA) and magnetic resonance angiogram (MRA) have been developed.5
For example, recent studies suggest high sensitivity and specificity for CTA in identifying intracranial aneurysms, approaching 95% and 100% respectively, when compared to the DCA (Figures 1 and 2).6 Yet DCA remains over-utilised, supported by the reported low rate of peri-procedural complications of only <1%.7 The current understanding of the risks associated with DCA and EP is based on ascertainment of clinical complications. Diffusion weighted (DW) MRI has emerged, in the last two decades, as an extremely sensitive tool for detecting acute ischemic lesions which uncommonly cannot be asymptomatic.8,9 DW MRI provides image contrast that is dependent on Brownian motion of water molecules, which may be altered by various neurological diseases. In acute ischemic stroke there is immediate energy depletion that leads to disruption of the neuronal cell membrane and ATP dependent sodium and potassium pump. This is accompanied by restriction in the motion of water molecules across the cell membrane. An abnormal signal in the DW MRI is indicative of irreversible neuronal damage and appears as abnormal hyper-intensity, occurring within half hour from the onset of acute ischemic stroke.
DW MRI has been exceedingly utilised both clinically and in stroke trials as the gold standard to detect new ischemic stroke lesions. Moreover, it has been shown as an ideal tool for quantifying ischemic injury related to EP.10 Since the introduction of this novel modality, numerous studies have been performed to evaluate the incidence of new ischemic lesions, as detected by the DW MRI, in patients undergoing EP for various therapeutic purposes, including carotid artery stent, aneurysm coiling, and AVM embolisation.11,12
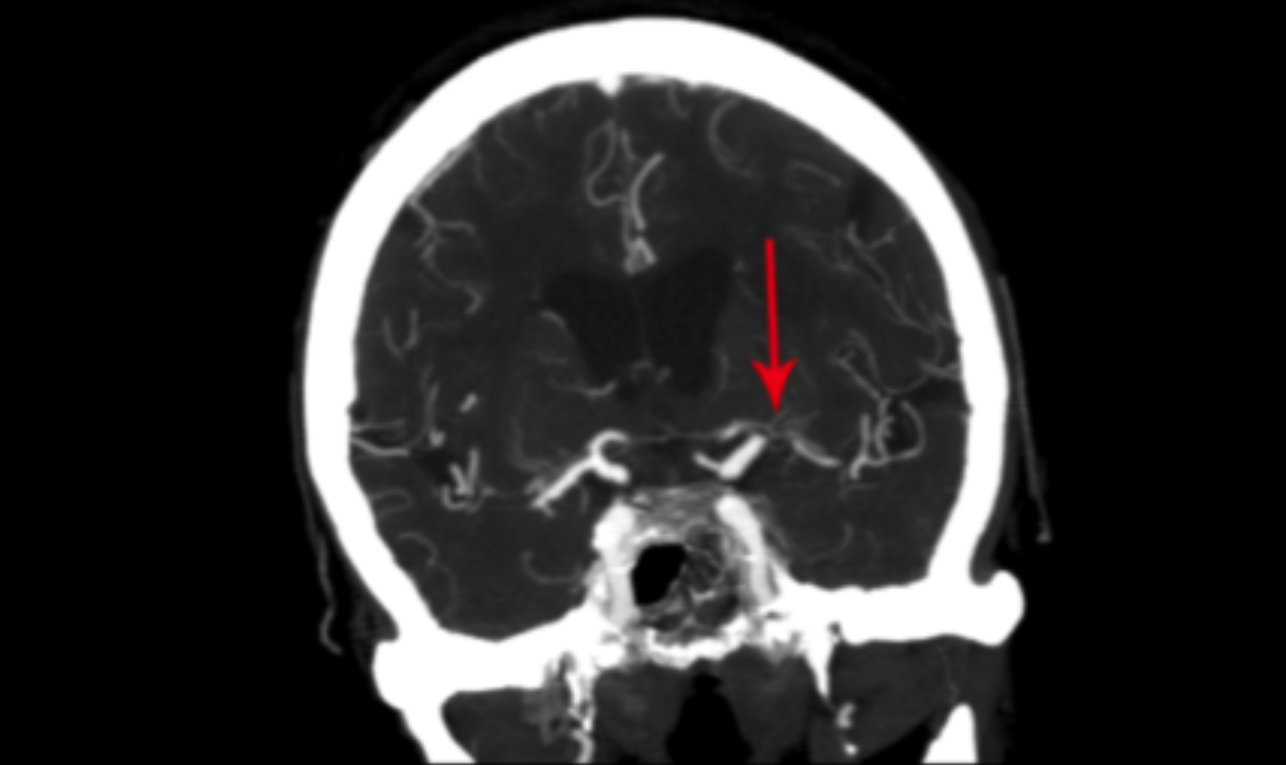 Figure 1: CT angiogram demonstrating left middle cerebral artery stenosis.
Figure 1: CT angiogram demonstrating left middle cerebral artery stenosis.
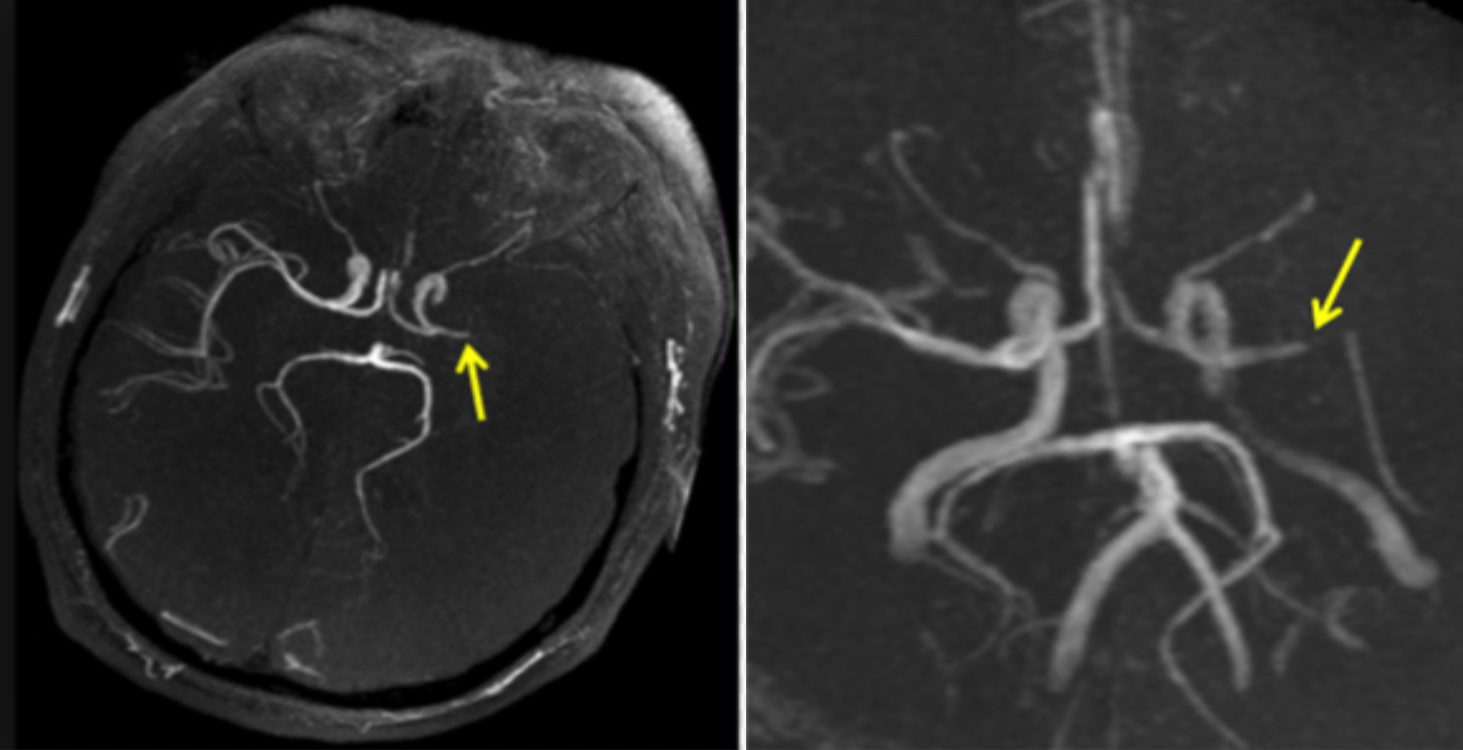 Figure 2: MR angiogram demonstrating left middle cerebral artery stenosis.
Figure 2: MR angiogram demonstrating left middle cerebral artery stenosis.
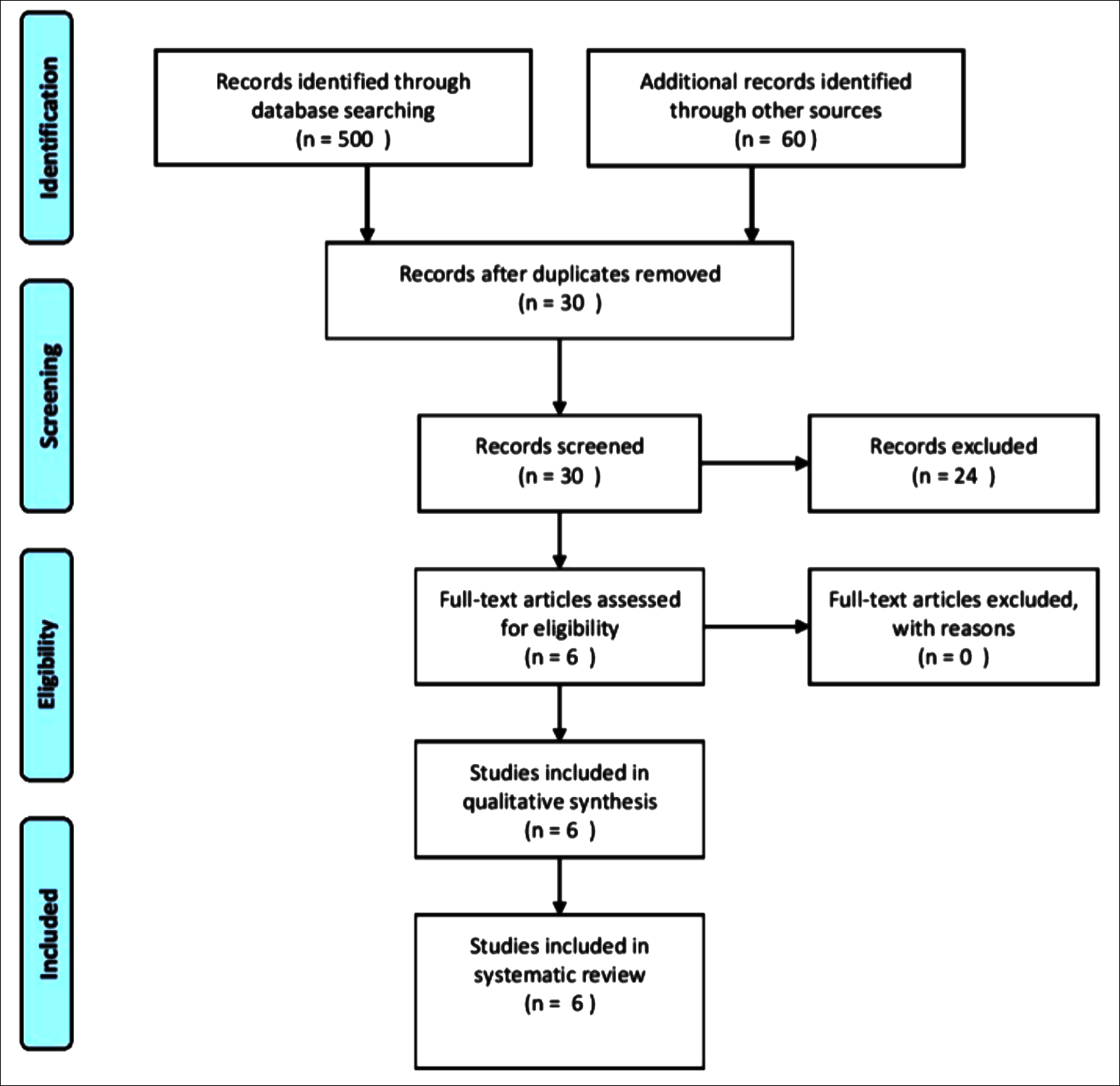 Figure 3: Prisma flow diagram.
Figure 3: Prisma flow diagram.
Several studies have also assessed the rate of new ischemic lesions by DW MRI in patients undergoing DCA. The rate of new ischemic lesions, based on the separate results of each of these studies, appears high; and frequently, the DWI abnormality does not even correspond to any obvious clinical symptoms or deficits. However, all of these studies were single-centre with relatively small sample size. Therefore, this systematic review was conducted to determine the risk of new ischemic lesions as evident on DW MRI, in patients undergoing DCA for various cerebrovascular purposes.
METHODOLOGY
Two academic neurologists searched the MEDLINE, EMBASE, Current Contents, Cochrane Central, Expert Files, and bibliographies of included articles to identify prospective studies that reported DW MRI findings after DCA. Key words for the search were a combination of stroke, ischemic event, angiogram, MRI, endovascular procedure, diffusion weighted MRI, and diagnostic angiogram. The search terms used in MEDLINE was: stroke, ischemic event, cerebral angiogram, and diffusion-weighted MRI. Five hundred and sixty studies were identified from all enlisted sources with no publication time limit. After filtering for duplicates, 30 articles were chosen for review. Two authors independently assessed all studies for eligibility, and finally six eligible studies were included in the systematic review. The PRISMA flow diagram for the search strategy is given in Figure 3.
Manuscripts written in English and published between 1999 and 2019, with prospective study design, where only DCA was carried out prior to the DWI MRI and pre-and post-procedure DW MRI (within one week prior to and after procedure) were included. The number of new ischemic lesions on DW MRI were documented after each procedure. Stroke with clinical manifestation within one week after the procedure was documented. Case studies, letters, editorials, and review articles were excluded. Data extracted from each manuscript included year of publication, number of endovascular procedures, purpose of procedure, number of new DW MRI lesions per each procedure, and incidence of clinically symptomatic new ischemic stroke after each procedure.
Results of all the included pooled studies were to estimate the rate of new ischemic lesions on DW MRI per 100 diagnostic angiogram procedures. Microsoft Excel 2013 was used for the statistical analysis in this review. Only qualitative statistics of the studies, such as number of participants, and number of DW MRI lesions were tabulated and presented. There was a considerable degree of heterogeneity among the studies, which prevented from making any quantitative synthesis in the form of meta-analysis. For the same reason, odds ratio, or pooled hazard ratios could not be used to create forest plots or subgroup analysis. Significance levels were presented, correspond to a p-value ≤0.05 as significant and denote the results obtained within each independent study only.
RESULTSA total of six studies met the eligibility criteria.13-18 Total number of diagnostic angiograms reported were 430. New DW MRI lesions were detected after 106 of the angiogram procedures (24.65%).
Table I: Characteristics of studies that met inclusion criteria.|
Name |
Year |
Number of patients |
DWI lesions |
Percentage |
|
Gossetti 18 |
2007 |
100 |
24 |
24.0% |
|
Bendszus 17 |
1999 |
66 |
23 |
34.8% |
|
Masayuki 16 |
2013 |
174 |
41 |
23.6% |
|
Kato 14 |
2003 |
50 |
13 |
26.0% |
|
Britt 13 |
2000 |
20 |
2 |
10.0% |
|
Chuah 15 |
2004 |
20 |
3 |
15.0% |
|
DWI: Diffusion-weighted imaging. |
||||
Around 11-17% of the procedures were associated with >1 new DW MRI lesions. The rate of new ischemic lesions among the included studies ranged from 10% to 34.8%. However, the incidence of new ischemic lesions increased proportional to the number of performed procedures (Table I). Factors identified to increase the risk for new DW MRI lesions after DCA included: fluoroscopy time of more than 17 minutes (OR 9.4, 95% CI 1.8 to 48.6, p=0.0078); utilising more contrast medium (149 vs. 110 ml, p <0.01); history of vascular risk factors (44% vs. 13%, p =0.03); additional catheters employed; vessels difficult to navigate; and procedures performed by resident operator (OR 2.526, 95% CI 1.214-5.254).
DISCUSSIONDCA continues to be frequently utilised in clinical medicine despite the advent and merit of less invasive procedures such as CT angiogram and MR angiography.6 However, the safety of angiogram is based on studies that rely on clinical symptoms and deficits as a measure of ischemic complications.7,19 DW MRI has emerged, in the last two decades, as the most sensitive tool for identifying acute ischemic lesions. Many of the DW MRI lesions, however, are clinically silent.20 However, there is ample evidence to suggest abnormal cognitive functions when a detailed neurocognitive assessment is exercised on these patients with silent DW lesions.18
DW MRI has been established as the gold standard tool for identifying new ischemic stroke. Hence, the safety of EP and DCA based on only clinical ascertainment of deficits is unreliable. DW MRI must be employed when assessing the safety of EP and DCA. Several studies based on single-centre design reported frequent rate of DWI lesions after EP.15-17
This systematic review, which included 430 diagnostic angiogram procedures, confirmed a high rate of new ischemic stroke after the procedure, approaching 24.65%. Moreover, the majority of the DW MRI abnormalities did not correspond to any clinical neurological symptoms or deficit. Mean time fluoroscopy was independently associated with increased risk for new ischemic lesion on MRI. Hence, angiogram operators must be vigilant in minimising fluoroscopy time. Interestingly, angiogram performed by residents in training was associated with increased risk for new DW MRI lesions. This can be probably attributed to increased procedure time and increased probing when the procedure is performed by inexperienced operator. This calls for a more careful and vigilant supervision by the instructor. It also calls for more utilisation of simulation-based angiogram training before allowing the residents to perform angiograms on real patients. Patients with multiple vascular risk factors were also found to be at increased risk of new ischemic lesions arising from diagnostic DCA. This can be explained by the increased prevalence of atherosclerotic plaques in this patient population.
The low number of procedures in two of the included studies,13,15 is a major limitation of this review. However, the high rate of DW MRI lesions in these two studies is consistent with the results of the other studies with much larger sample size.
DCA continues to be frequently and unjustifiably employed despite the high sensitivity and specificity of non-invasive procedures such as CT angiogram approaching in some instances 95% and 100%, respectively.6 The postulated mechanisms responsible for the new ischemic lesions after the procedure, include the release of atherosclerotic plaque through manipulation of catheter at the proximal large arteries such as the aorta and brachiocephalic artery; spontaneous thrombus formation at the catheter; and thrombus formation triggered by injury of the endothelium of the large arteries.
The alarming results of this systematic review must warn clinicians against the lenient and hasty use of DCA, unless deemed absolutely necessary. Instead, non-invasive procedures must be encouraged for diagnostic purposes; especially now that the reliability of non-invasive procedures, in terms of sensitivity and specificity, is rapidly, steadily, and progressively improving. Hence, it is expected that the quality of data generated from these procedures will match or even supersede the quality generated by the conventional angiogram. The results of this study should not preclude physicians from using DCA; but it should alarm them of the frequent rate of stroke complications, associated with the procedure, so that it is utilised only when deemed necessary.
CONCLUSION
DCA have been utilised for the diagnosis of many cerebrovascular conditions. While it has been typically associated with a low rate of clinical complications. We report a significantly high rate of 24.65% for new ischemic lesions, detected on DW MRI after DCA. Although most of the new lesions were found asymptomatic, complete neurological assessment would likely reveal neurocognitive abnormalities, which can be clinically significant. Care must be exercised in advising DCA. Unless absolutely necessary, alternative procedures such as CT angiography and MR angiography should be resorted to. For patients undergoing DCA, mean time fluoroscopy should be minimised; vigilant resident supervision should be encouraged; and special care must be taken in patients with pre-existing vascular disorders.
FUNDING DISCLOSURE:
This study was funded by the Deanship of Scientific Research at King Saud University through Research Group No. RGP-1438-008.
CONFLICT OF INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
FAH: Had the initial idea and contributed in the writing proposal and manuscript.
BA, AA: Contributed in search for relevant trials, using special search words and also their quality assessment.
YM: Contributed in writing manuscript and abstract.
REFERENCES
- Kyu HH, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, et al. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: A systematic analysis for the global burden of disease study 2017. Lancet 2018; 392(10159):1859-922. doi: 10.1016/S0140-6736(18)32335-3.
- Komotar RJ, Mocco J, Wilson DA, Connolly ES, Lavine SD, Meyers PM. The natural history of intracranial carotid artery atherosclerosis. Neurosurgical focus 2005; 18(1):1-3.
- Barnett HJM, Taylor DW, Haynes RB, Sackett DL, Peerless SJ, Ferguson GG, et al. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Eng J Med 1991; 325(7):445-53. doi: 10.1056/ NEJM199108153250701.
- Chicoine MR. Microsurgery and clipping: The gold standard for the treatment of intracranial aneurysms. J Neurosurgical Anesthesiol 2003; 15(1):61-3. doi: 10.1097/00008506- 200301000-00013.
- Schellinger PD, Richter G, Köhrmann M, Dörfler A. Noninvasive angiography (magnetic resonance and computed tomography) in the diagnosis of ischemic cerebrovascular disease. Cerebrovascular Dis 2007; 24(Suppl. 1):16-23. doi: 10.1159/000107375.
- Dehdashti AR, Rufenacht DA, Delavelle J, Reverdin A, De Tribolet N. Therapeutic decision and management of aneurysmal subarachnoid haemorrhage based on computed tomographic angiography. Br J Neurosurg 2003; 17(1): 46-53.
- Heiserman JE, Dean BL, Hodak JA, Flom RA, Bird CR, Drayer BP, Fram EK. Neurologic complications of cerebral angiography. Am J Neuroradiol 1994; 15(8):1401-7.
- Warach S, Chien D, Li W, Ronthal M, Edelman RR. Fast magnetic resonance diffusion‐weighted imaging of acute human stroke. Neurol 1992; 42(9):1717. doi: 10.1212/wnl.42.9.1717.
- Warach S, Gaa J, Siewert B, Wielopolski P, Edelman RR. Acute human stroke studied by whole brain echo planar diffusion‐weighted magnetic resonance imaging. Ann Neurol 1995; 37(2):231-41. doi: 10.1002/ana.410370214.
- Fiebach JB, Schellinger PD, Jansen O, Meyer M, Wilde P, Bender J, et al. CT and diffusion-weighted MR imaging in randomised order: Diffusion-weighted imaging results in higher accuracy and lower interrater variability in the diagnosis of hyperacute ischemic stroke. Stroke 2002; 33(9):2206-10. doi: 10.1161/01.str.0000026864.20339.cb.
- Schnaudigel S, Gröschel K, Pilgram SM, Kastrup A. New brain lesions after carotid stenting versus carotid endarterectomy: a systematic review of the literature. Stroke 2008; 39(6):1911-9. doi: 10.1161/STROKEAHA.107.500603.
- Shibazaki K, Iguchi Y, Kimura K, Ueno Y, Inoue T. New asymptomatic ischemic lesions on diffusion-weighted imaging after cerebral angiography. J Neurol Sci 2008; 266(1-2):150-5. doi: 10.1016/j.jns.2007.09.017.
- Britt PM, Heiserman JE, Snider RM, Shill HA, Bird CR, Wallace RC. Incidence of postangiographic abnormalities revealed by diffusion-weighted MR imaging. Am J Neuroradiol 2000; 21(1):55-9.
- Kato K, Tomura N, Takahashi S, Sakuma I, Watarai J. Ischemic lesions related to cerebral angiography: Evaluation by diffusion weighted MR imaging. Neuroradiol 2003; 45(1): 39-43. doi: 10.1007/s00234-002-0889-5.
- Chuah KC, Stuckey SL, Berman IG. Silent embolism in diagnostic cerebral angiography: Detection with diffusion‐weighted imaging. Australasian Radiol 2004; 48(2):133-8. doi: 10.1111/j.1440-1673.2004.01273.x.
- Sato M, Nakai Y, Tsurushima H, Shiigai M, Masumoto T, Matsumura A. Risk factors of ischemic lesions related to cerebral angiography and neuro-interventional procedures. Neurologia medico-chirurgica 2013; 53(6):381-7. doi: 10.2176/nmc.53.381.
- Bendszus M, Koltzenburg M, Burger R, Warmuth-Metz M, Hofmann E, Solymosi L. Silent embolism in diagnostic cerebral angiography and neurointerventional procedures: A prospective study. Lancet 1999; 354(9190):1594-7. doi: 10.1016/S0140-6736(99)07083-X.
- Gossetti B, Gattuso R, Irace L, Faccenna F, Venosi S, Bozzao L, et al. Embolism to the brain during carotid stenting and surgery. Acta chirurgica Belgica 2007; 107(2):151-4.
- Dion JE, Gates PC, Fox AJ, Barnett HJ, Blom RJ. Clinical events following neuroangiography: A prospective study. Stroke 1987; 18(6):997-1004. doi: 10.1161/01.str.18.6.997.
- Schofer J, Arendt M, Tübler T, Sandstede J, Schlüter M. Late cerebral embolization after emboli-protected carotid artery stenting assessed by sequential diffusion-weighted magnetic resonance imaging. JACC: Cardiovascular Interventions. 2008; 1(5):571-7. doi: 10.1016/j.jcin.2008.06.008.