MicroRNA-16, Programmed Cell Death Protein-4 (PDCD-4) and Polycystic Ovarian Syndrome
By Naseem Zehra, Arfa Azhar, Rehana RehmanAffiliations
doi: 10.29271/jcpsp.2020.08.880Sir,
Polycystic ovarian syndrome (PCOS) is a prevalent disease in 7-9% of the women of reproductive age.1 The severity of PCOS can be determined by Rotterdam criteria, depending upon the spectrum of characteristics present, including hyperandrogenism (H), and/or lack of ovulation (O), and/or ovarian cysts (OC). These include category-1: H+O, category-2: O+OC, category-3: H+OC, and category-4: all 3 characteristics present. Its reproductive disturbances manifest as menstrual irregularities, lack of ovulation, impaired follicular maturation, hyperandrogenism, which eventually funnel to infertility. Its metabolic disturbances, present in almost half of PCOS patients,2 include high blood pressure, insulin resistance, obesity and abnormal lipid profile.1
Micro-RNAs have single strands and small segments of non-coding RNA (18-25 nucleotides), which break down RNA and prevent translation by binding to 3’-UTR at post-transcriptional level, thus are important players in cell growth and death.1,3 Studies focusing on altered serum miRNA levels in PCOS, suggest its use as a prognostic and diagnostic marker for PCOS.2
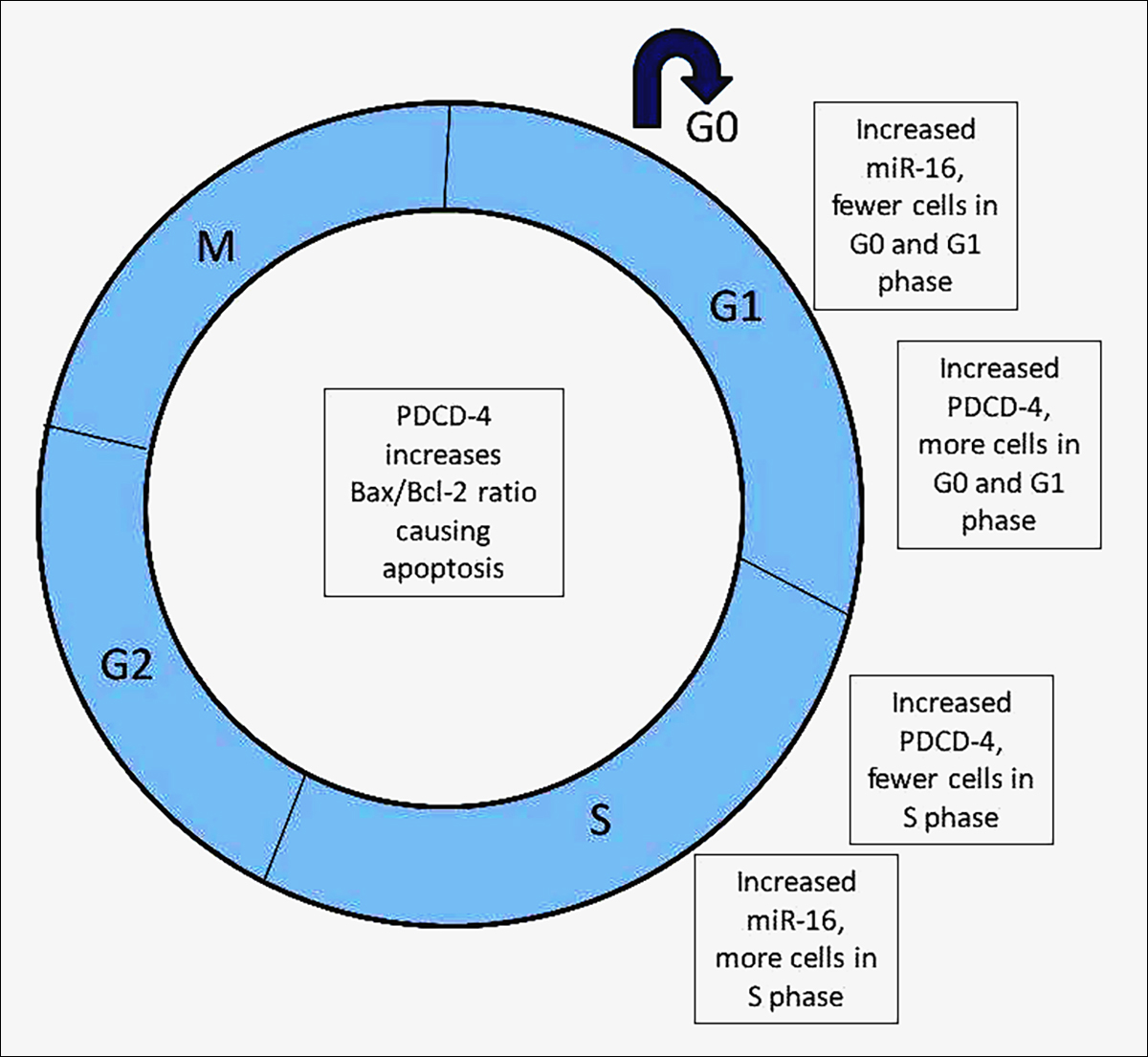 Figure 1: Diagrammatic representation of effect of MiRNA-16 and PDCD-4 on cell cycle.4
Figure 1: Diagrammatic representation of effect of MiRNA-16 and PDCD-4 on cell cycle.4
The association between miR-16 and programmed cell death protein-4 (PDCD-4) and their contributory roles in the progression of PCOS is still under analytic study. PDCD-4, present on chromosome 10q24, increases in PCOS and induces apoptosis by down regulating RNA helicase activity during translation. MiR-16, on chromosome 13q14, is low in PCOS. Fu et al. stated that the level of miR-16 in the ovarian cortical cells and blood was decreased in PCOS, while ovarian cortex’s PDCD-4 level was increased.2 Contrarily, increased miR-16 in PCOS women’ granulosa cells promoted cellular proliferation and decreased cell death, where a greater number of cells were in the S phase and few in GO-G1 phase, while increased PDCD-4 and decreased miR-16 expression reversed these effects. High testosterone levels in PCOS alter folliculogenesis by decreasing miR-16 and increasing PDCD-4, resulting in more cell death and decreased cellular proliferation.1 Thus, it can be concluded that the impaired follicular growth and granulosa cell death in PCOS can be improved by increasing miR-16 expression which has an effect on PDCD-4 at the molecular level.1
MiRNA-16 levels can then serve as a confirmatory diagnostic test for PCOS. Thus, a sensitive and specific diagnosis can be carried out which is minimally invasive, less expensive and comfortable for the patients, unlike transvaginal ultrasonography.2
It shows isolated effects of MiRNA-16 and PDCD-4 on different stages of the cell cycle. Different levels of miRNA-16 and PDCD-4 change the balance of cells, particularly in the GO, G1 and S phase. High levels of PDCD-4 result in a positive impact and an increase in the number of cells in the G0 and G1 phase, while cause an opposite effect on the cells in S phase, i.e. a decrease in their number. On the other hand, increased serum levels of MiRNA-16 have opposite effects on the cells in the 3 phases. These findings agree with the findings of the cellular proliferative and anti-apoptotic effect of MiRNA-16, as fewer cells will be in the resting stage and more will be in the DNA replicative stage, leading to the mitotically growing stage.
CONFLICT OF INTEREST:
Authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
NZ: Conceived the idea and did literature search.
AA: Helped in literature search and write-up.
RR: Reviewed the write-up.
REFERENCES
- Fu X, He Y, Wang X, Peng D, Chen X, Li X, et al. Microrna-16 promotes ovarian granulosa cell proliferation and suppresses apoptosis through targeting pdcd4 in polycystic ovarian syndrome. Cell Physiol Biochem 2018; 48(2):670-82.
- Bhatnager R, Nanda S, Dang AS. Plasma prolidase levels as a biomarker for polycystic ovary syndrome. Biomark Med 2018; 12(6):597-606.
- Sorensen AE, Wissing ML, Englund AL, Dalgaard LT. Microrna species in follicular fluid associating with polycystic ovary syndrome and related intermediary phenotypes. J Clin Endocrinol Metab 2016; 101(4): 1579-89.
- Ding L, Gao F, Zhang M, Yan W, Tang R, Zhang C, et al. Higher PDCD 4 expression is associated with obesity, insulin resistance, lipid metabolism disorders, and granulosa cell apoptosis in polycystic ovary syndrome. Fertil Steril 2016; 105(5):1330-7.e3.